Pharmaceutical Quality (Industry), published by HL7 International / Biomedical Research and Regulation. This guide is not an authorized publication; it is the continuous build for version 1.0.0 built by the FHIR (HL7® FHIR® Standard) CI Build. This version is based on the current content of https://github.com/HL7/uv-dx-pq/ and changes regularly. See the Directory of published versions
Provide details about drug substance and drug product components, materials, and Quality reference standards.
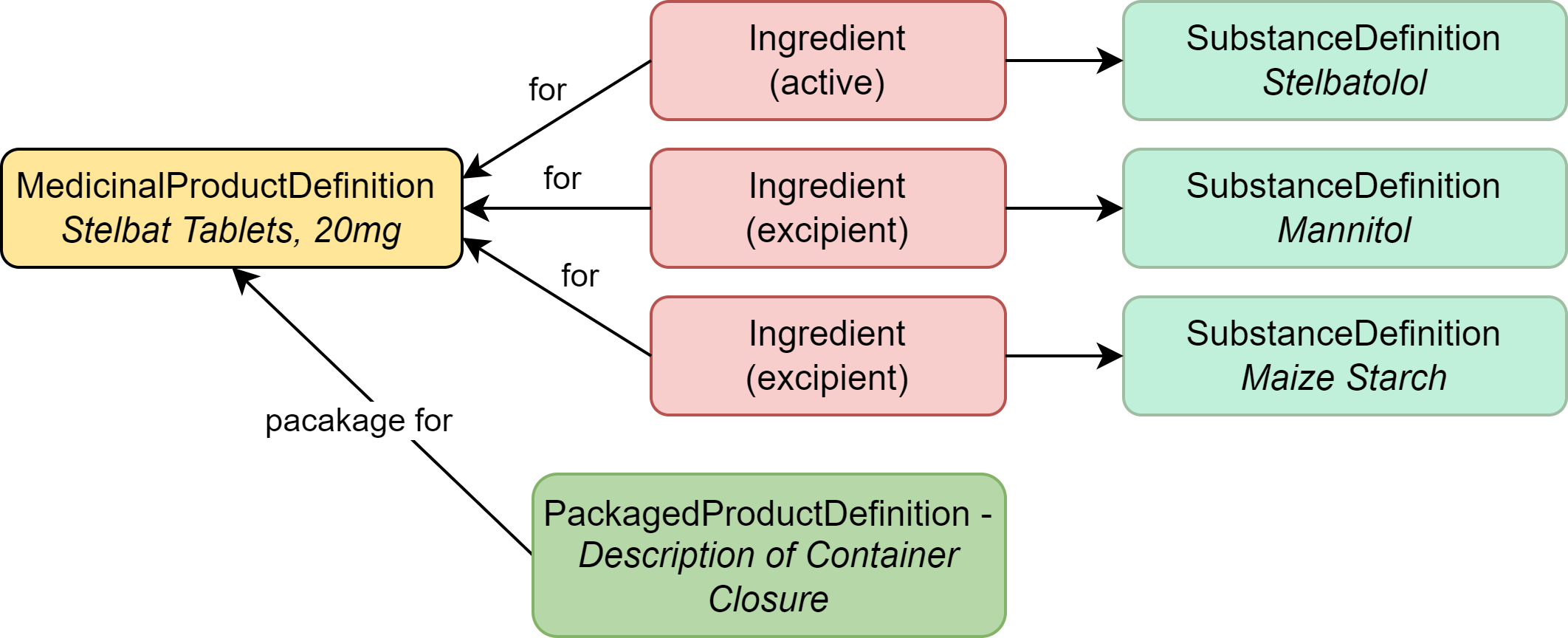 |
| MedicinalProductDefinition | The drug product (Stelbat tablets, 20mg) |
| Ingredient | The active ingredient (stelbatalol) and other ingredients that make up the product |
| SubstanceDefinition | The documented impurities |
| PackagedProductDefinition | Information about the packaging for the product |
CTD section synthetic source data samples (PDF):
XML and JSON examples of synthetic quality data:
HTML presentation example of synthetic quality data:
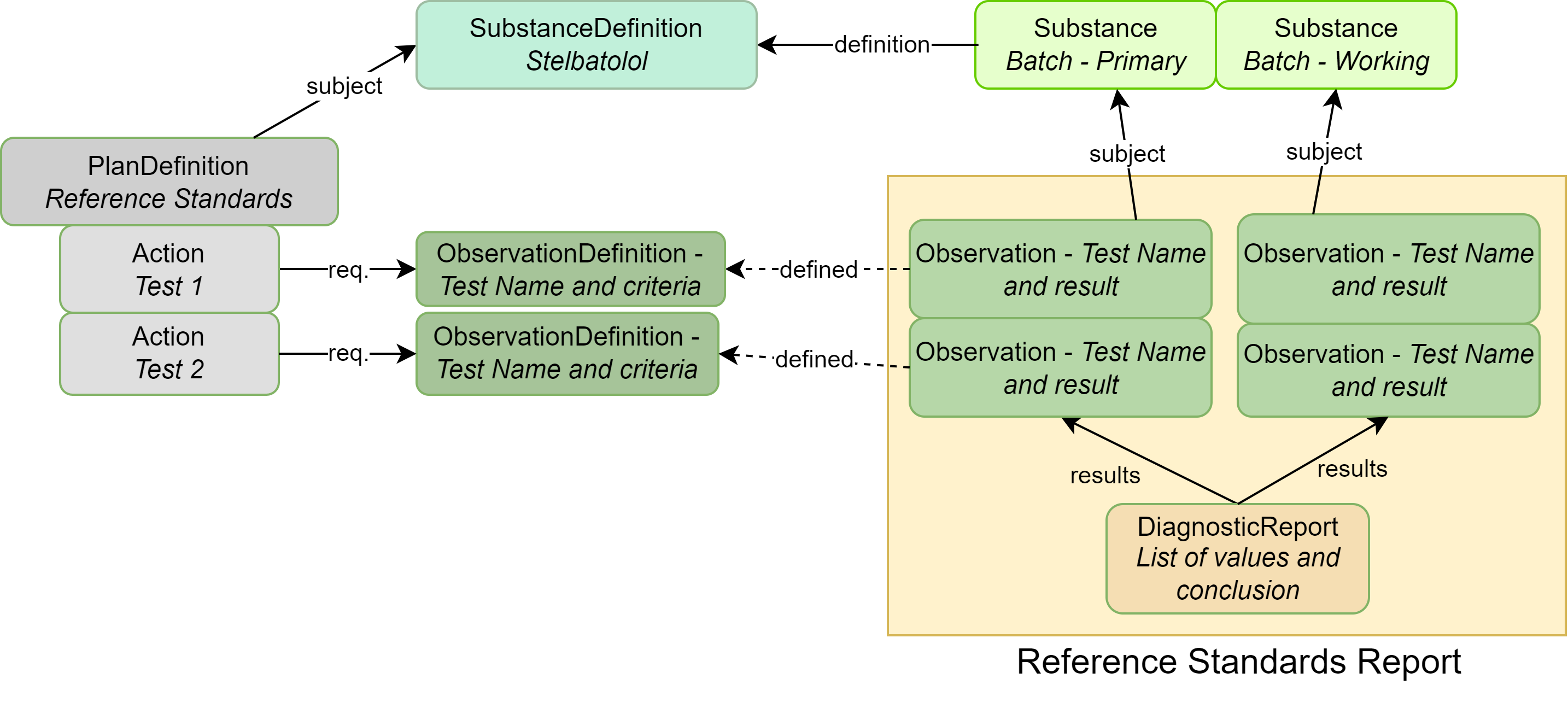 |
| SubstanceDefinition | The substance for which reference standards are being established |
| Substance | Identifying information about an actual batch of substance (an instance) |
| PlanDefinition | Describes the reference standards set of tests |
| ObservationDefinition | Each individual test and acceptance criteria |
| Observation | The results of a specific test mentioned in the ObservationDefinition |
| DiagnosticReport | Contains all results as a group and captures conclusions |
| Organization | (not illustrated above): The company/site that performed the testing or manufacturing |
CTD section synthetic source data samples (PDF):
XML and JSON examples of synthetic quality data:
HTML presentation example of synthetic quality data: