xShare Project IPS+
0.1.0 - qa-preview
150
xShare Project IPS+
0.1.0 - qa-preview
150
xShare Project IPS+, published by xShare Project. This guide is not an authorized publication; it is the continuous build for version 0.1.0 built by the FHIR (HL7® FHIR® Standard) CI Build. This version is based on the current content of https://github.com/hl7-eu/xshare-ips-plus/ and changes regularly. See the Directory of published versions
The data domain describes the data model and the data exchange format.
The main purpose of the IPS+ content is connecting health and healthcare to research and public health. When one thinks of research and public health, one thinks of CDISC suite of standards which is why the mapping of the data elements goes to the CDISC standards which support rigorous clinical trials and are required for submissions to regulatory bodies. The OMOP common data model, and HL7 FHIR have content as well; however, the use cases are different. OMOP provides the signals of what is happening in the world; whereas CDISC provides the structure and rigor CDISC required to test the hypothesis of what scientists would like to verify. FHIR provides the exchange standards for communicating between stakeholders. The key connector is the data element concept.
The guide includes data element concepts common across health, research and public health and maps them. The maps provide the NIH NCIt C-codes that link to the CDISC biomedical concepts thus enabling end users to pull in the entire picture (all pertinent variables surrounding the concept to fully inform the question and answer). This is needed to test the hypothesis through the observational data. CDISC standards support end –to-end research and the tools within this guide are meant to connect HL7 FHIR, CDISC standards and the OMOP common data model with guidance for the CDISC terminology, and SNOMED. LOINC is used where appropriate. This linkage uses the core harmonized data element set.
The figure below provides a view of the data elements as recommended in the Proposal for a harmonized core data set across health care, population health and clinical research for the content of the IPS+. These recommendations are the results of:
The data element concepts are aligned with the CDISC Domain and the equivalent IPS Category.

Adverse Events (AEs) in prospective studies are new conditions after trial enrolment, and would likely be collected in EMR/EHR/PHR systems as Conditions. Their classification as AEs is due to the timing relative to study enrolment.
These concepts are central to IPS+ in the context of interventional clinical trials (e.g., treatment efficacy and safety). The collection of AEs and Serious Adverse Events (SAEs) ensures participant safety.
An SAE may include any of the following:
In the context of a drug/device trial, a formal SAE report must assess the impact of the study on the event and guide protective actions.
The SAE report includes:
All these components are included in the IPS+ data model. Automating SAE population in an eCRF via European EHRxF would greatly improve safety workflows.
If new occurrences of conditions or allergies arise in the patient, even during a clinical trial, these data would also be entered in an EHR as new conditions and allergies using its usual data elements. Therefore, in terms of mapping, these additional data element concepts specified for Adverse Events will likely be documented only on the research side and the full Adverse Event data element set does not need to be added to the IPS. Thus, the only additional concepts not in the current IPS are Research Subject, Research Study and the Healthcare Encounter data element set.
To simplify data mapping in PHRs, several CDISC resources can be used:
These tools support transformation from FHIR IPS to the CDISC SDTM specification.
FHIR-CDISC mapping documents and aligned value sets aid in enabling secondary data use from PHRs.
Note that CDISC uses MedDRA codes, whereas the IPS is assumed to use SNOMED CT. A SNOMED CT to MedDRA mapping is maintained and published by SNOMED International. Leveraging the FHIR to CDISC maps, the value sets provided that are aligned with the CDISC terminology will assist in providing support for PHRs that can be leveraged for secondary data use.
The illustrations show how the data element concepts connect across the supporting data element items. The key topic variable is the main data element that without it, the rest of the data element items are meaningless. The below figure shows the question and the answers on the case report form for medical history. The table on the right shows the medical history term (condition) and the surrounding related variables (supplemental qualifiers) that would be found in the biomedical concept for medical history (Medical History Reported Term), bc id C83119 which is the same as the nci code of C83118. The figure shows how SNOMED terminology represents the question of “what is the clinical finding?” and the answer of “diabetes mellitus” (with the corresponding SNOMED codes).

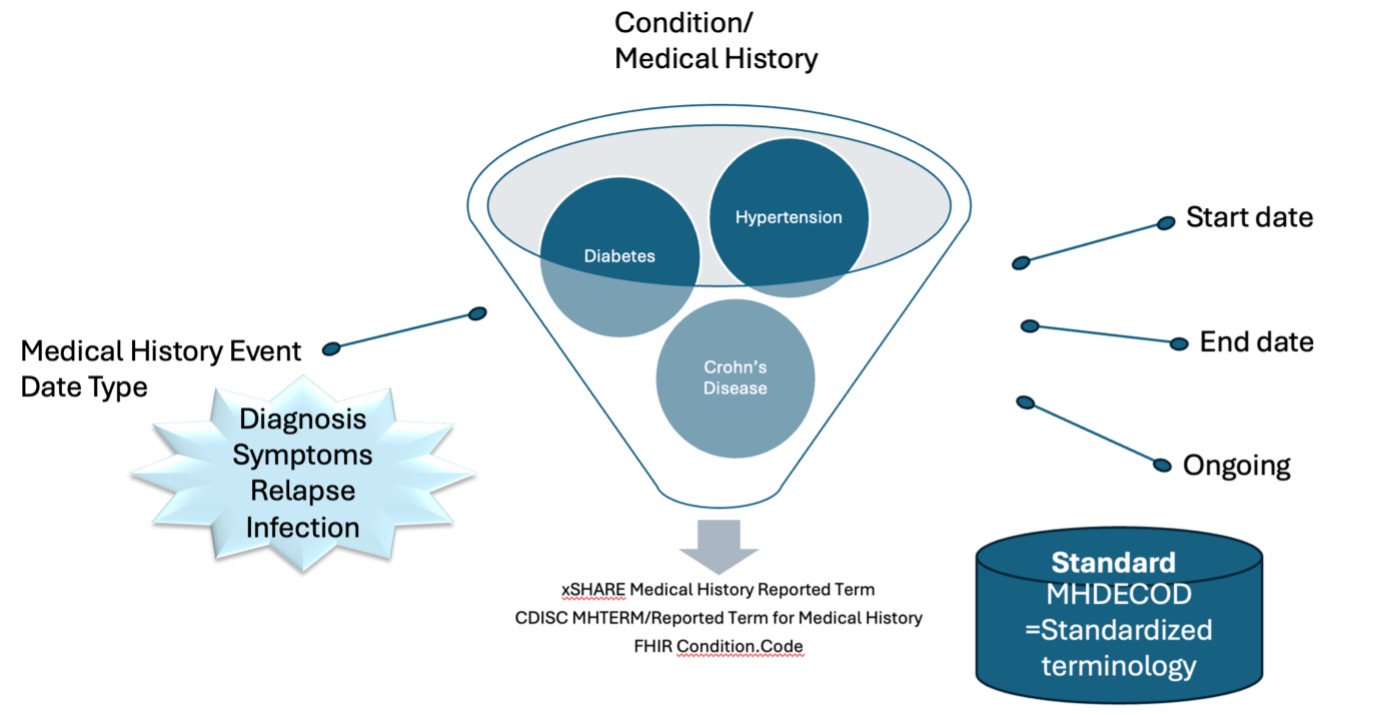
Below figure is another view of how the data element concepts are related. The funnel as the connector by concepts and terminology and the surrounding concepts as the qualifying data element items that support the data element concepts. In CDISC terms, the Reported Term for the Medical History (MHTERM) with the synonym qualifier of the Dictionary-Derived Term (MHDECOD), the timing variables of the Start Date/Time of the Medical History Event and the End Date/Time of the Medical History Event. The Medical History Event Date Type (MHEVDTYP) is a qualifier used to represent the aspect of the medical condition or event by which MHSTDTC and/or the MHENDTC is defined. Examples: "DIAGNOSIS", "SYMPTOMS", "RELAPSE", "INFECTION". In clinical research, the question is often asked if the event is ongoing which is a CDASHIG variable, Ongoing Medical History Event (MHONGO). The terminology provides the traceability across the CDISC suite of standards as well as across standard to standard.