
http://loinc.org
http://snomed.info/sct
http://unstats.un.org/unsd/methods/m49/m49.htm
urn:iso:std:iso:3166
urn:iso:std:iso:3166:-2
This fragment is available on en/licenses.html
This publication includes IP covered under the following statements.
| Type | Reference | Content |
|---|---|---|
| web | somelocation | signatureUrl: https://somelocation |
| web | github.com | Clinical Study Protocol, published by HL7 International / Biomedical Research and Regulation. This guide is not an authorized publication; it is the continuous build for version 1.0.0-ballot2 built by the FHIR (HL7® FHIR® Standard) CI Build. This version is based on the current content of https://github.com/HL7/vulcan-udp-ig/ and changes regularly. See the Directory of published versions |
| web | www.HL7Vulcan.org |

|
| web | github.com | Support is available at the DDF GitHub . |
| web | github.com | For more examples and also to find links a viewer for this content go to the Vulcan UDP Examples GitHub page |
| web | github.com | The relationships between the elements of M11, USDM and FHIR are shown in the Mapping spreadsheet described in the following section. The present focus of this mapping is representation of M11 so the mapping does not cover the whole of USDM. There is a useful infographic from CDISC here: ( usdm_m11_classes ) that shows the overlap between M11 and USDM and in the bottom right USDM classes not covered by M11. |
| web | hl7vulcan.org | Why Vulcan ? |
| web | github.com | NOTE: The relationship between the M11 protocol template specification and the structured and unstructured capabilities of the USDM is illustrated on the DDF GitHub site. Each logical area of the M11 protocol template is highlighted along with the associated area within the USDM. See infographic usdm_m11.pdf |
| web | www.cdisc.org | USDM is a reference architecture produced by CDISC to be used by TransCelerate and " anyone who wishes to create conformant standards-based technologies, which produce or consume study definitions - source DDF-RA/README.md at v4.0.0 · cdisc-org/DDF-RA · GitHub ". |
| web | www.transceleratebiopharmainc.com | USDM is a reference architecture produced by CDISC to be used by TransCelerate and " anyone who wishes to create conformant standards-based technologies, which produce or consume study definitions - source DDF-RA/README.md at v4.0.0 · cdisc-org/DDF-RA · GitHub ". |
| web | github.com | USDM is a reference architecture produced by CDISC to be used by TransCelerate and " anyone who wishes to create conformant standards-based technologies, which produce or consume study definitions - source DDF-RA/README.md at v4.0.0 · cdisc-org/DDF-RA · GitHub ". |
| web | github.com | Content based on DDF-RA (GitHub) used under the CC-BY-4.0 license. |
| web | creativecommons.org | Content based on DDF-RA (GitHub) used under the CC-BY-4.0 license. |
| web | github.com | The Implementation Guide makes NO changes to this content with the exception of copying content from the m11 mapping document . The actual mappings are not changed. |
| web | database.ich.org | M11 Template |
| web | database.ich.org | M11 Guideline |
| web | database.ich.org | M11 Technical Specification |
| web | github.com | CDISC USDM Data Model |
| web | www.ema.europa.eu | ICH M11 (EMA) |
| web | www.cdisc.org | CDISC Digital Data Flow (DDF) |
| web | www.hl7vulcan.org | HL7 Vulcan Utilizing the Digital Protocol |
| web | euclinicaltrials.eu | CTIS |
| web | github.com | CTIS Mapping |
| web | github.com | SDTM Mapping |
| web | github.com | ClinicalTrials.gov Mapping |
| web | github.com | Common Protocol Template Mapping |
| web | github.com | M11 Mapping |
| web | github.com | Enterprise Vocabulary Services](https://evs.nci.nih.gov/tools). Within the options presented there are packages for POSTMAN™ that allow the querying of the thesaurus. For the preparation of this IG we used the FHIR R5 set. |
../assets/images/001.svg 
|
Extensions and profiles 02.png 
|
FHIR-components.png 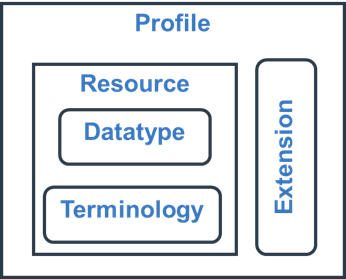
|
Mapping.png 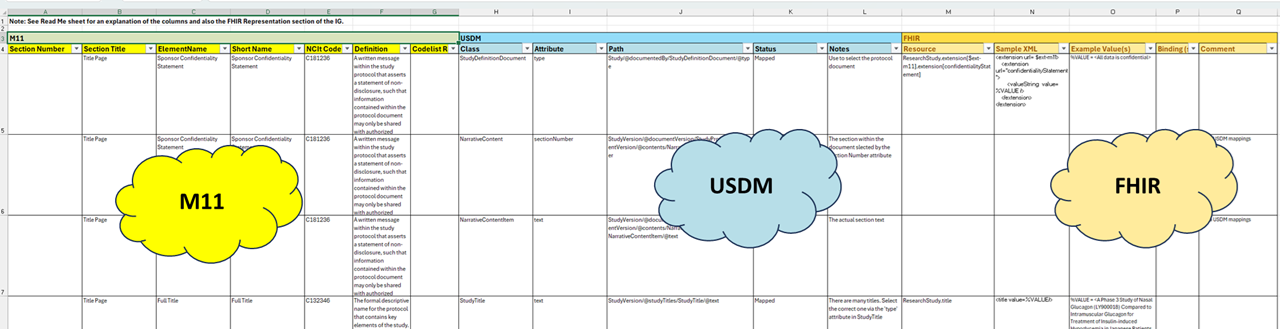
|
bundles-01.png 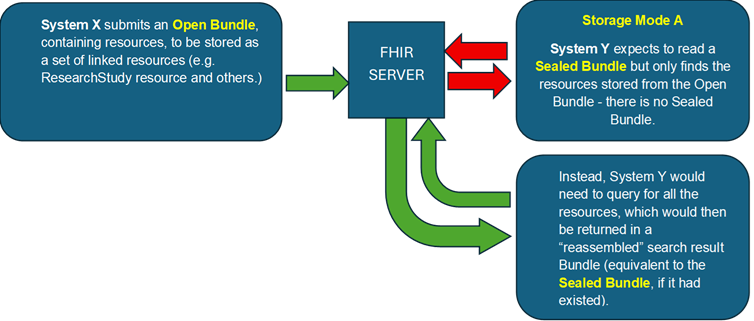
|
bundles-02.png 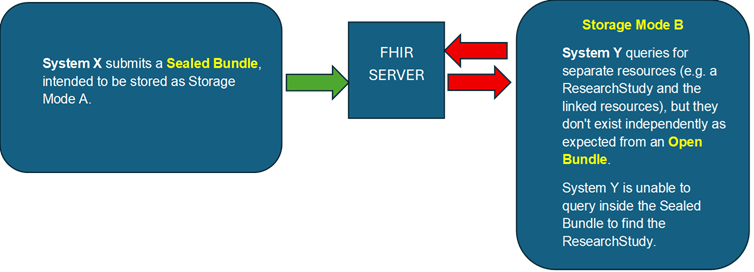
|
iceberg-03.png 
|
image-20260129155349453.png 
|
image-20260129155431823.png 
|
image-20260129164106045.png 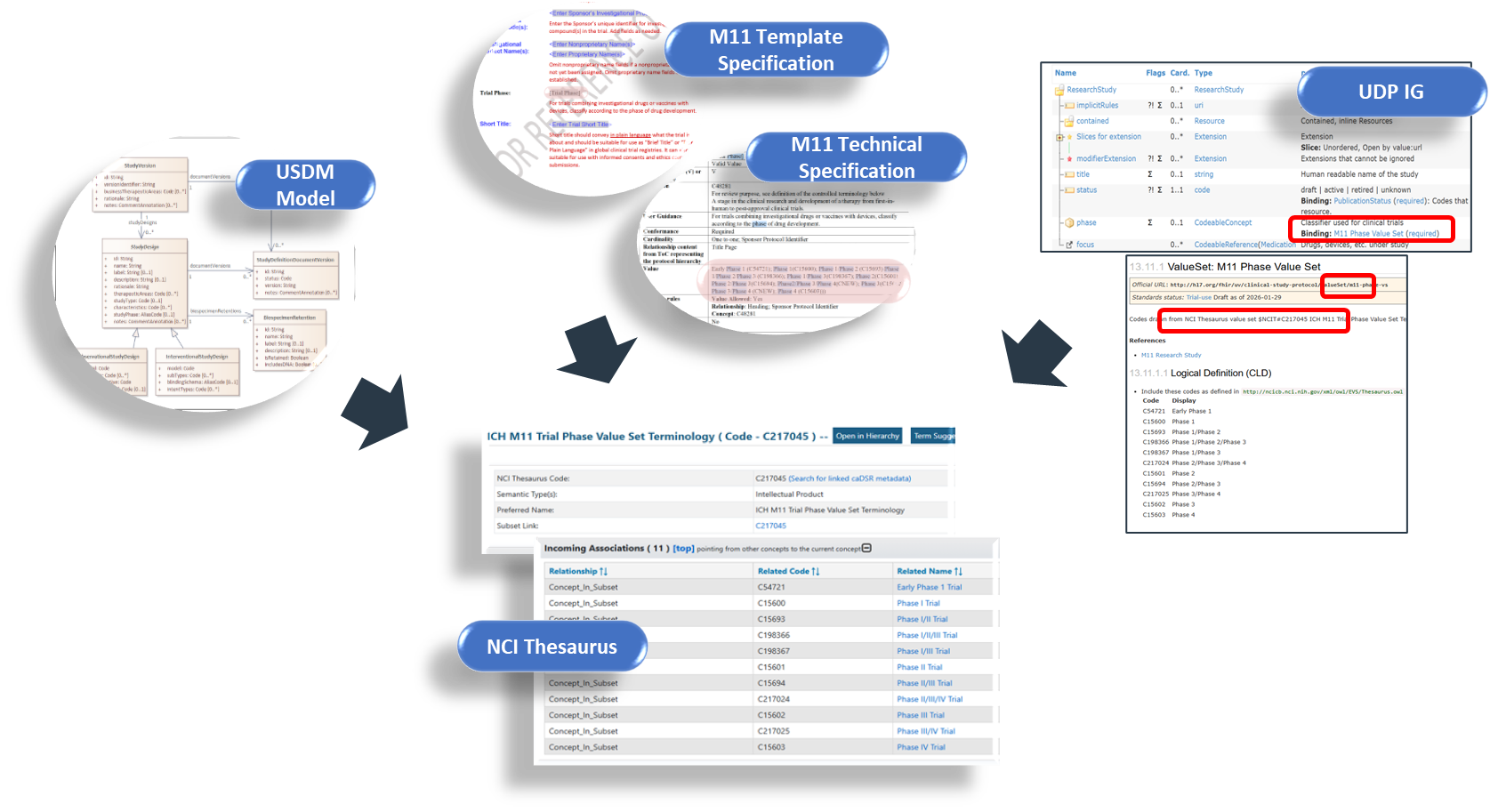
|
image-20260129165903655.png 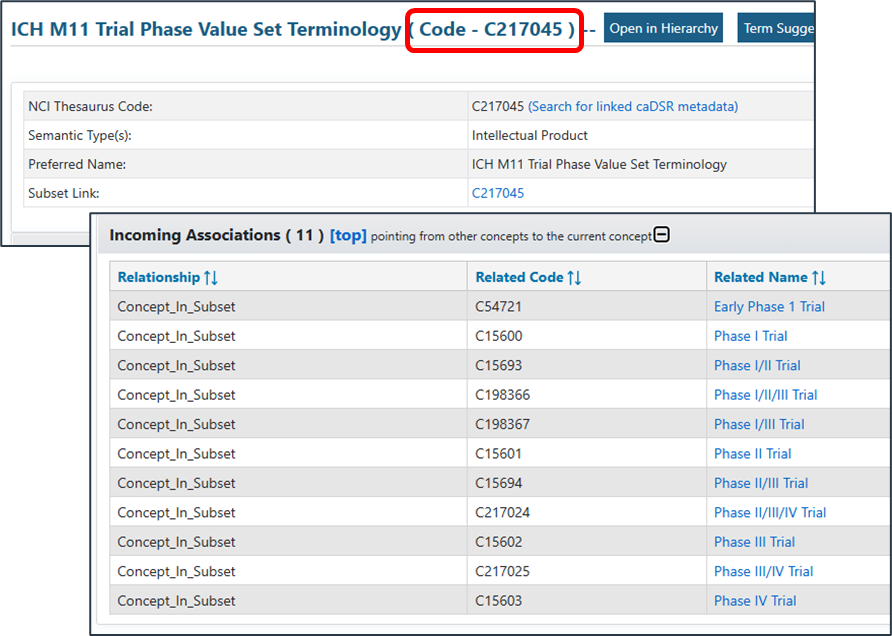
|
image-20260129165955355.png 
|
image-20260129170010341.png 
|
narrative-and-structured-02.png 
|
narrative-content-01.png 
|
narrative-content-02.png 
|
section-vs-01.png 
|
shipping-01.png 
|
structured-content-01.png 
|
tree-filter.png 
|
work-flow-protocol-approval.png 
|