Clinical Study Protocol, published by HL7 International / Biomedical Research and Regulation. This guide is not an authorized publication; it is the continuous build for version 1.0.0-ballot2 built by the FHIR (HL7® FHIR® Standard) CI Build. This version is based on the current content of https://github.com/HL7/vulcan-udp-ig/ and changes regularly. See the Directory of published versions
| Page standards status: Informative |
Selected use cases of value across clinical research and health IT settings can be enabled through FHIR-based exchange of narrative protocol text and structured protocol content.
The detail looks first at the use case addressed in development of this Implementation Guide, then looks towards additional use cases with which compatibility will be necessary and at future opportunities.
Validation: An Implementation Guide can specify required data elements, be prescriptive about the format of data items, specify the terminology coded items must be drawn from and check the data meets constrtaints specified in the implementation guide. This is done my importing the machine readable form of the implementation guide into the server and the transmitted data stating that it meets the restrictions of the implementation guide.
Thus the role of a FHIR server in automatically validating content according to the requirements of an implementation guide should be considered within the workflow. This allows both the sender and the receiver to validate content according to their chosen Implementation Guide. Validation by the sender and then again by the receiver gives both parties confidence in the quality of the data being transmitted.
For the purposes of development of this initial version of this Implementation Guide, the use case was defined narrowly as enabling sponsor-to-regulator exchange of digital protocols aligned to ICH M11 in FHIR. The specific detailed technical and business requirements for submission of M11-aligned protocols to regulators will be determined as regulators adopt M11 and establish processes for receipt and utilization of digitized protocols. Because this simplistic approach was taken, it is expected that this Implementation Guide could also be utilized by other sender/receiver roles.
The processes supporting testing scenarios were articulated as simply and with as much flexibility for implementation as possible:
Ensure protocol content and organization aligned to ICH M11
Generate protocol in FHIR
Send protocol
Receive protocol in FHIR
Validate receipt of protocol (i.e., display, extraction of structured content)
NOTE: methods for generation, exchange, and validation of receipt will vary. The Vulcan UDP project welcomes experience from stakeholders.
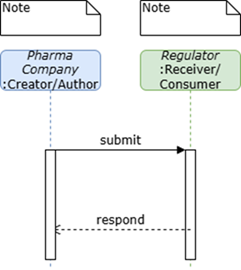
Figure 1: Sponsor to Regulator Use Case
In order to ensure this Implementation Guide is set up to enable compatibility for future use cases and future specificity of requirements, variations on the above simple process were considered, such as:
The information contained in a protocol is utilized in relation to numerous processes across the research and care continuum. Digitizing and enabling interoperable exchange of that information opens an opportunity for automation, reuse, improved quality across stakeholders.
The Vulcan UDP project intends to address additional use cases. All are welcome to propose use cases to Vulcan and to participate in Vulcan UDP community meetings.
Vulcan UDP is not the only group seeking to leverage digital protocols. Many have been mentioned within this Implementation Guide and others exist as well. In alignment with Vulcan principles, the Vulcan UDP project aims to ensure compatibility, avoid duplication, and make use of collective resources whenever possible. Coordination with other groups will be essential to this aim.
To get a sense of the extent of use cases that exist across the ecosystem, see the visual below. Digitization of existing processes are often the first use cases implementers want to achieve, but the possibilities go beyond. Many of the use cases at the “tip of the iceberg” as well as those “below the surface” are currently implemented within certain environments though perhaps using bespoke approaches and models. Increasing compatibility across artifacts (i.e., standards, guidelines) will help to accelerate and broaden the value of digital protocols across many use cases.
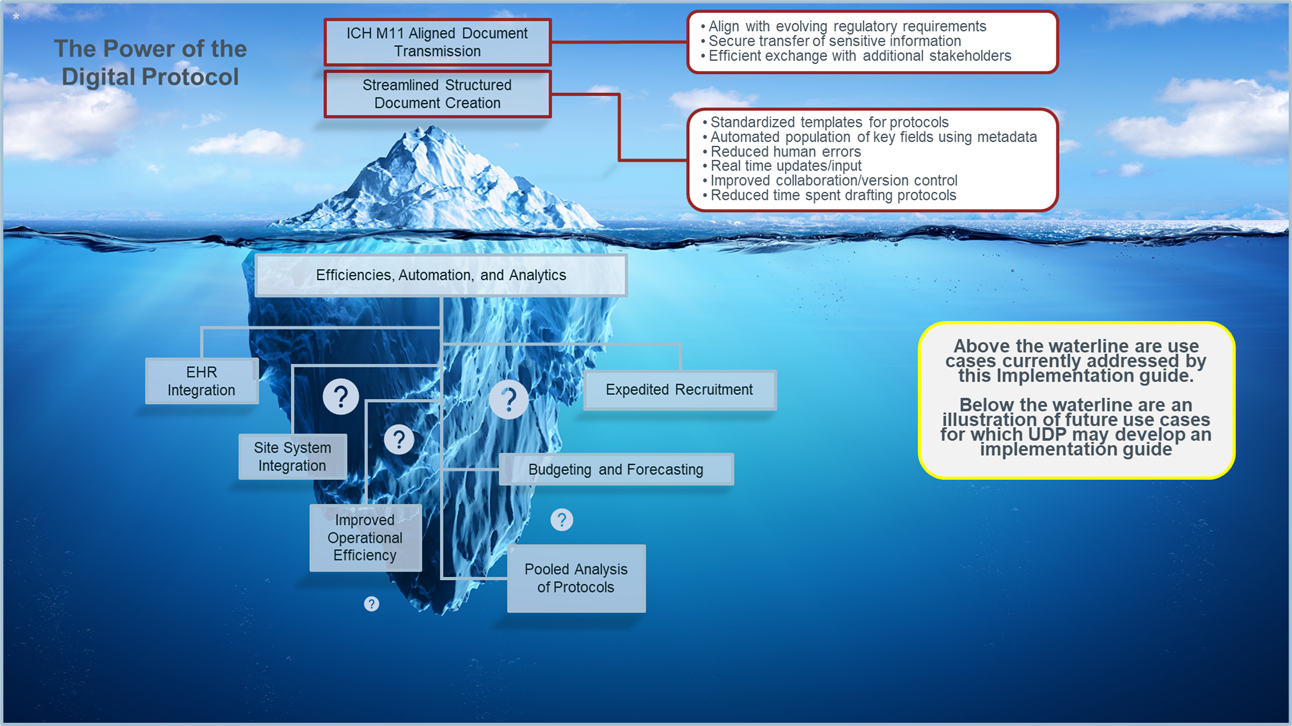
Figure 2: Example Future Use Cases