HL7 Europe Laboratory Report, published by HL7 Europe. This guide is not an authorized publication; it is the continuous build for version 2.0.0 built by the FHIR (HL7® FHIR® Standard) CI Build. This version is based on the current content of https://github.com/hl7-eu/laboratory/ and changes regularly. See the Directory of published versions
| Official URL: http://hl7.eu/fhir/laboratory/ImplementationGuide/hl7.fhir.eu.laboratory | Version: 2.0.0 | |||
| Active as of 2026-05-05 | Computable Name: Hl7EuLaboratoryIg | |||
Copyright/Legal: Copyright HL7 Europe. Licensed under creative commons public domain (CC0 1.0). |
||||

Clinical laboratory results play an important role in diagnosis, treatment, and follow-up of patients. The availability of high quality test results, and the capacity of sharing them, is therefore essential being often the basis for clinical decision making. For this reason the Laboratory has been selected as one of the priority domains for the European EHR eXchange Format (E-EHRxF).
Specify a set of rules to be applied to HL7 FHIR to define how to represent a Laboratory Report in the European Context, coherently with the European eHN Guidelines (see the European eHealth - Key documents ).
This Implementation Guide applies to laboratory reports within the core fields of in-vitro diagnostics, for example clinical biochemistry, haematology, immunohematology, microbiology, immunology, while leaving out some specialised laboratory domains like histopathology or medical genetics. This version focuses only on common rules that apply to all the in-scope situations, without specifying specialized domain-specific profiles, as for example microbiology profiles.
This guide is not limited to test results performed by clinical laboratories on Human specimens (from human subject), but it considers also results on non-human materials or living subjects; or non-human specimens paired with a human subject. Derived guides may restrict the scope as needed (e.g. limiting the scope to well-identified human beings)
The goal of this Implementation Guide is to define an European standard for the Laboratory Report to facilitate the harmonization among the national initiatives and prepare the ground for the European EHR eXchange Format (E-EHRxF).
This project is promoted by HL7 Europe, but realized in collaboration with several other European and national organizations and projects.
The aspiration of this guide is that of being used as basis for European National Guides, the European EHRxF and - consequently - by MyHealth@EU for the EU cross-border services.
The project background is described in the background page.
The solution adopted by this guide - and detailed in the Design choices page - balances the business requirement of Laboratory Report as legally signable document (i.e. as a FHIR document), with the expectation to get Lab Report by searching per DiagnosticReport. All this, taking into account the R5 DiagnosticReport design pattern where the DiagnosticReport <-> Composition relationship is directed from the DiagnosticReport to the Composition resource. This is done by supporting both perspectives (see figure below) requiring the document bundle ( BundleLabReportEu ) to always include a DiagnosticReport ( DiagnosticReportLabEu ) and enabling the pre-adoption of the R5 rules for the inclusion of entries in the Document Bundle.
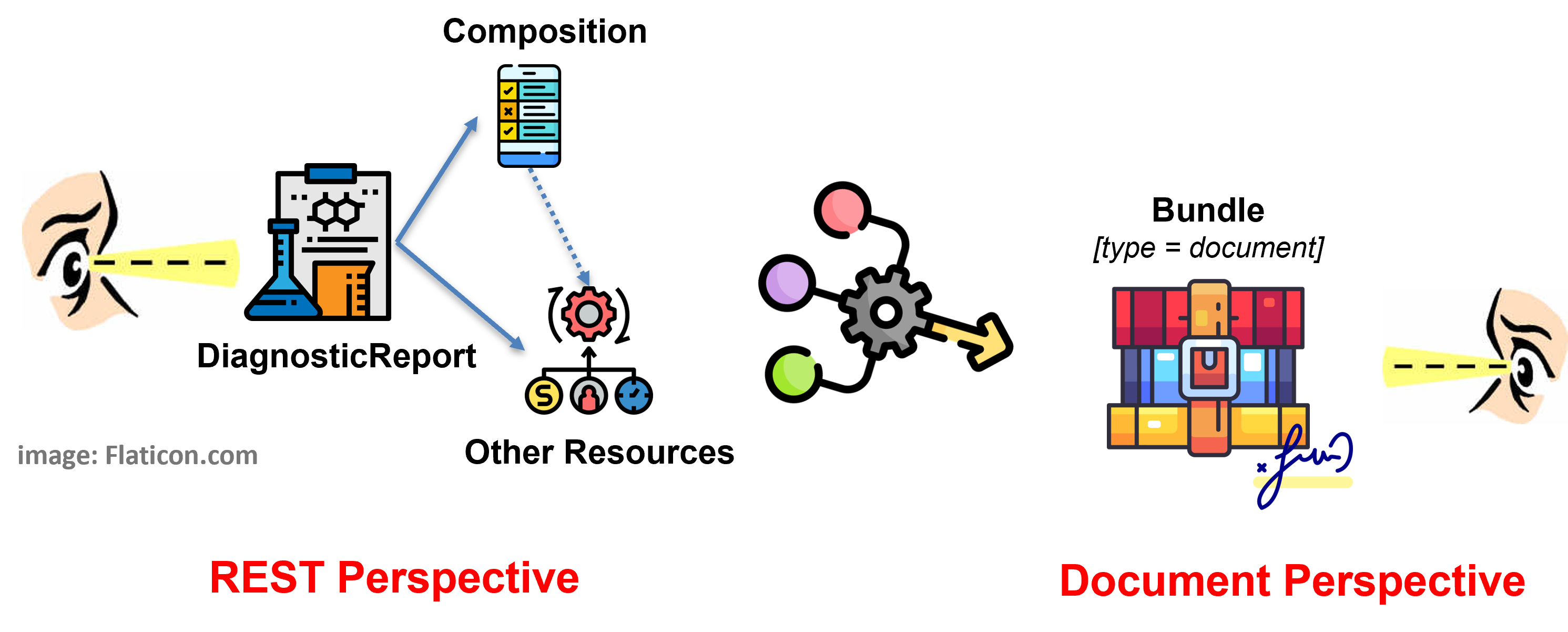
Figure 1 - Overview of the report design approach
The following diagrams provide a browseable overview of the profiles specified by this guide (not all the relationships have been reported).
The first highlights the most relevant relationships starting from the DiagnosticReport ( DiagnosticReportLabEu ) resource (REST Perspective).
Figure 2 - Overview of the profiles relationships
The second the profiles included in the document bundle ( BundleLabReportEu ) (Document Perspective).
Figure 3 - Overview of the document structure
Please refer to the Authors and Contributors page.