Opioid Prescribing Support Implementation Guide
2016.4.0 - CI Build
Opioid Prescribing Support Implementation Guide, published by Centers for Disease Control and Prevention (CDC). This guide is not an authorized publication; it is the continuous build for version 2016.4.0 built by the FHIR (HL7® FHIR® Standard) CI Build. This version is based on the current content of https://github.com/cqframework/opioid-cds-r4/ and changes regularly. See the Directory of published versions
Before starting opioid therapy for chronic pain, clinicians should establish treatment goals with all patients, including realistic goals for pain and function, and should consider how opioid therapy will be discontinued if benefits do not outweigh risks. Clinicians should continue opioid therapy only if there is clinically meaningful improvement in pain and function that outweighs risks to patient safety (recommendation category: A, evidence type: 4).
Contents
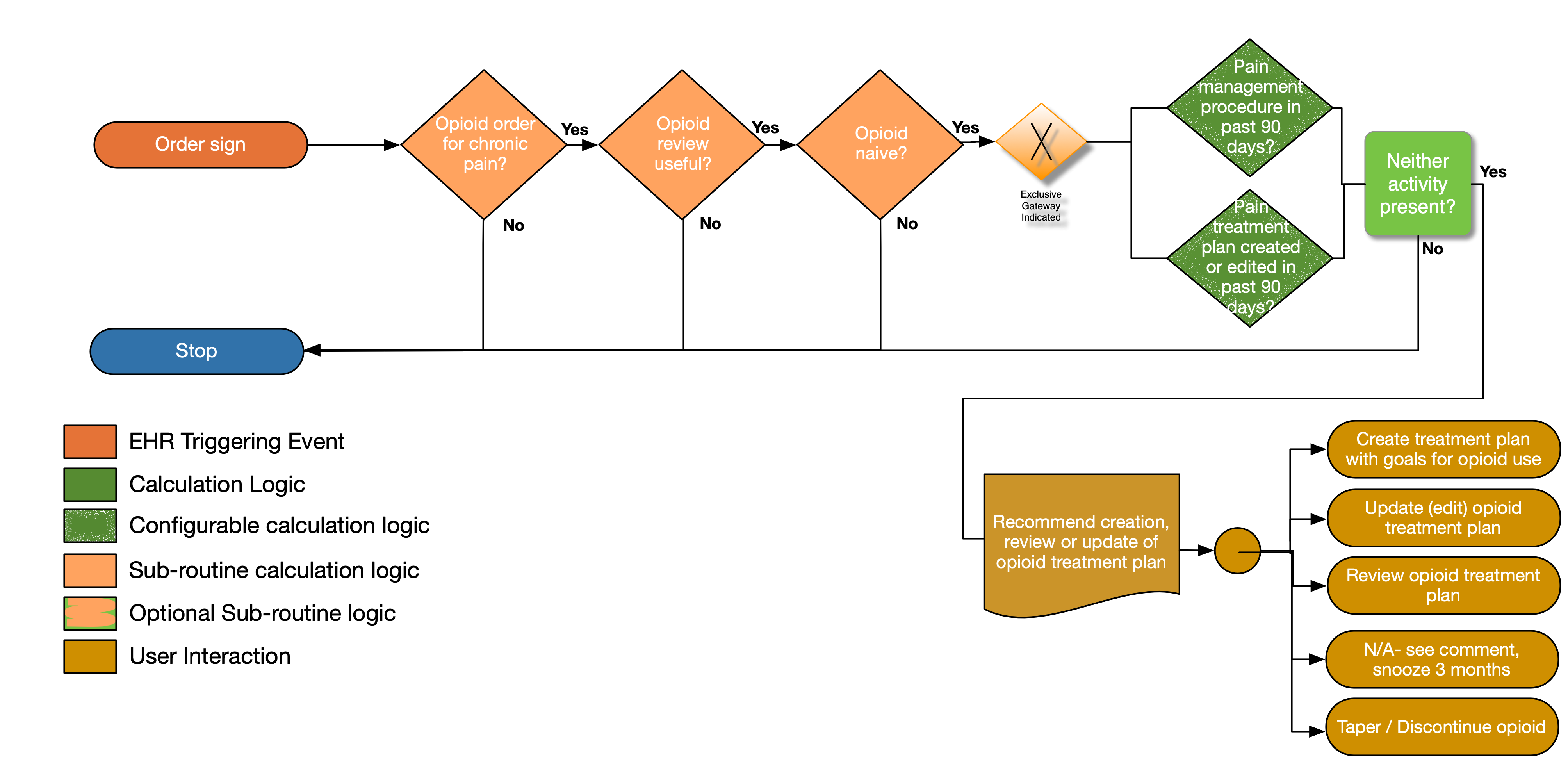
| Definition | Answer to Proceed | Details | Data (Terminology) Requirement |
|---|---|---|---|
| Opioid order for chronic pain? | Yes | See For Chronic Pain sub-routine | |
| Opioid review useful? | Yes | See Opioid Review Useful sub-routine | |
| Opioid naïve? | Yes | See Opioid Naïve sub-routine | |
| Pain treatment plan created or edited in past 90 days? | No | Look for a pain treatment plan that has been established or edited within the past 90 days (including the day of the visit). The following criteria should be met: • the treatment plan has an initiation date or an edited date within the past 90 days • the treatment plan should have been created or reviewed within the past 90 days by the same individual who is writing the new prescription |
Pain treatment plan |
| Pain management procedure in past 90 days? | No | • Look for a pain management procedure (including the procedure to review a pain management treatment plan that occurred within the past 90 days (including the day of the visit). | Pain management procedure |
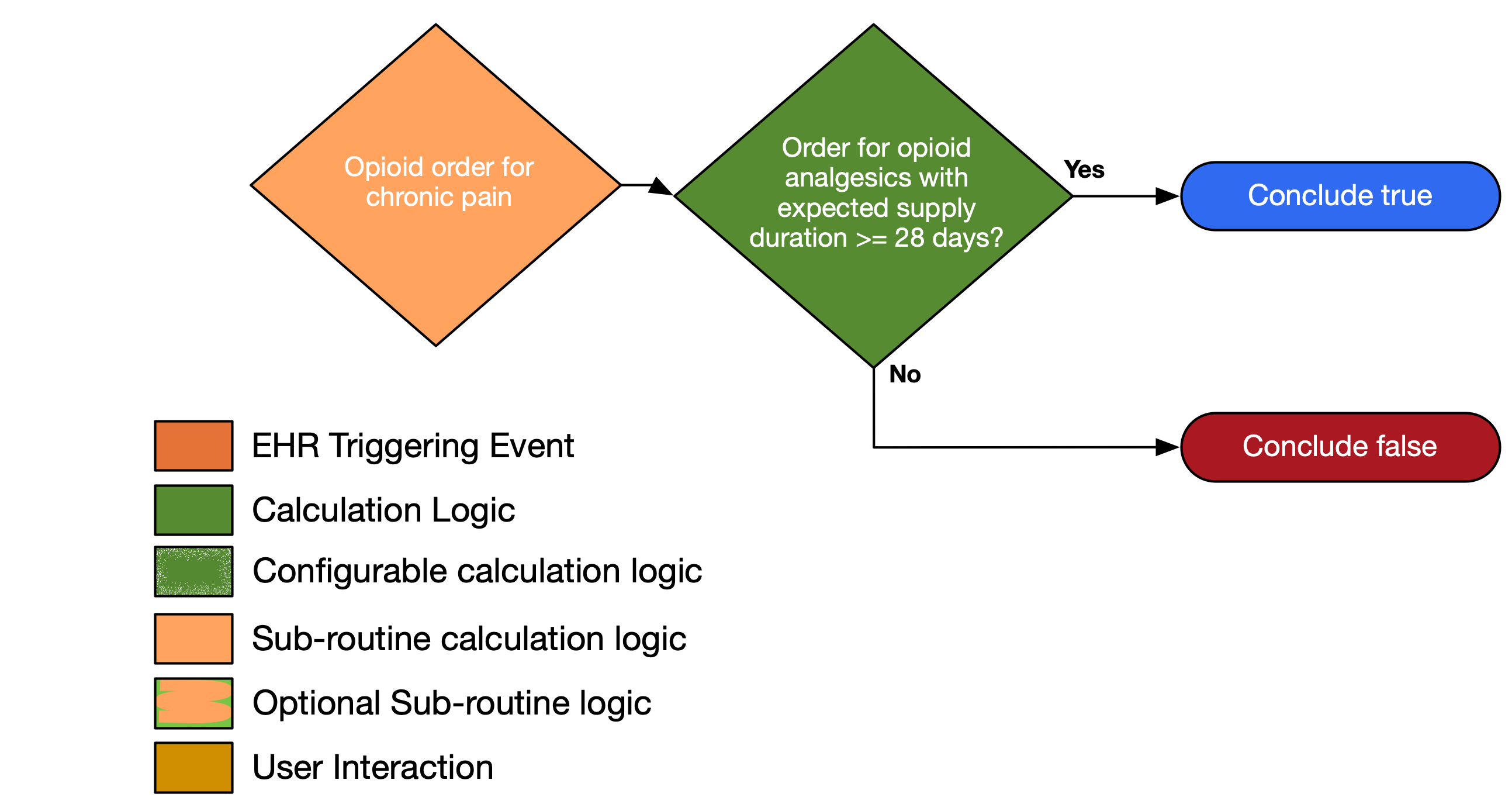
| Definition | Answer to Proceed | Details | Data (Terminology) Requirement |
|---|---|---|---|
| Order for opioid analgesic with expected supply duration ≥ 28 days | Yes | Order for opioid analgesics with ambulatory misuse potential with a supply duration of ≥ 28 days | Opioid analgesics with ambulatory misuse potential |
| Definition | Answer to Proceed | Details | Data (Terminology) Requirement |
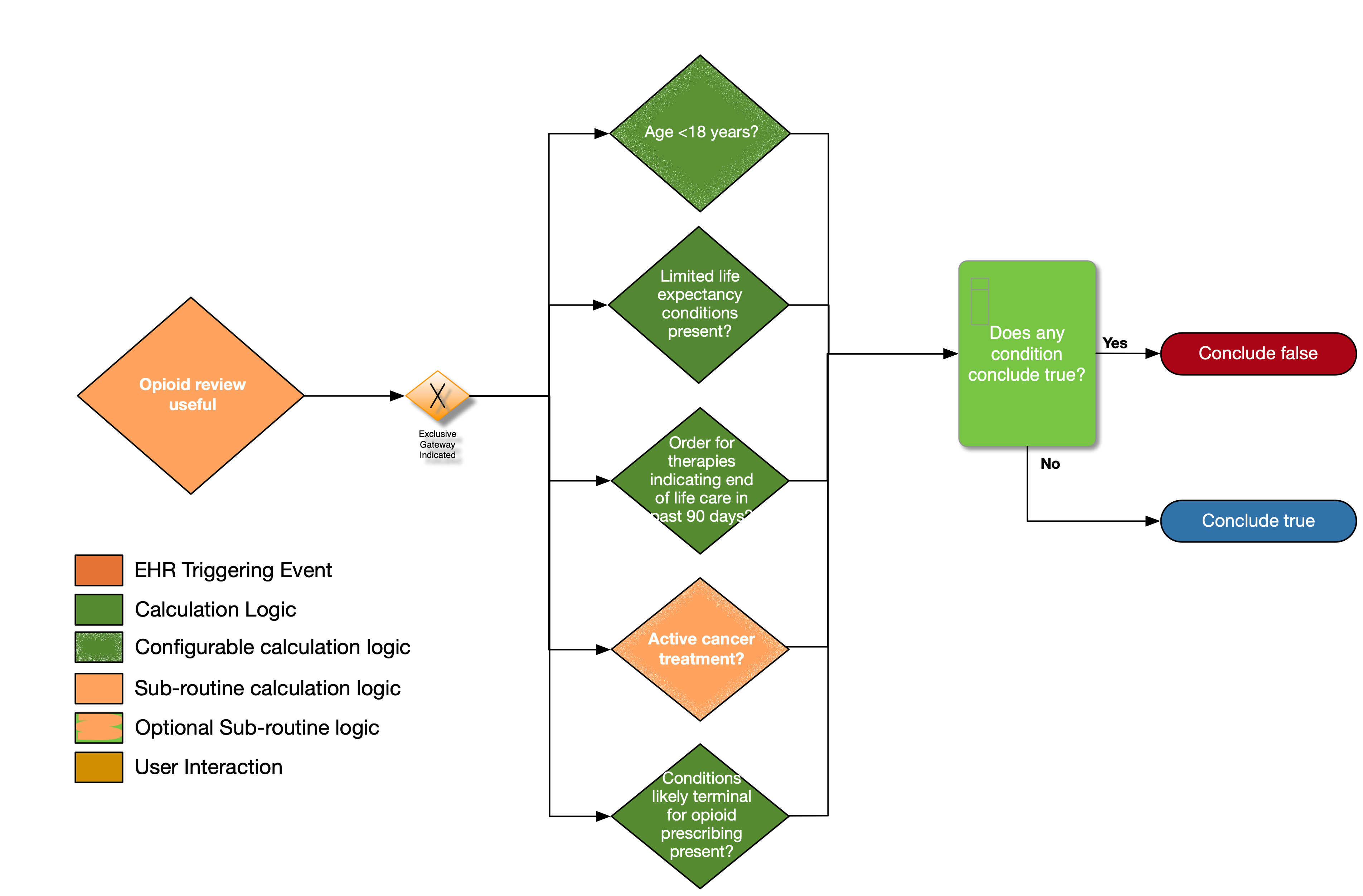
|---|---|---|---|
| Age < 18 years? | No | Calculate age from date of birth; exclude patients with age less than 18 years at the time of the prescription | Date of birth |
| Limited life expectancy conditions present? | No | Look for documented findings consistent with those listed in the limited life expectancy value set (terminal illness, bad prognosis, pre-terminal) | Limited life expectancy conditions |
| Order for therapies indicating end of life care in past 90 days? | No | Look for patients with an existing order for therapies indicating end of life care written within past 90 days | Therapies indicating end of life care |
| Active cancer treatment? | No | See Active Cancer Treatment sub-routine | See Active Cancer Treatment sub-routine |
| Conditions Likely Terminal for opioid prescribing present? | No | Look for patients with active conditions in the value set end-of-life-conditions | Conditions likely terminal for opioid prescribing |
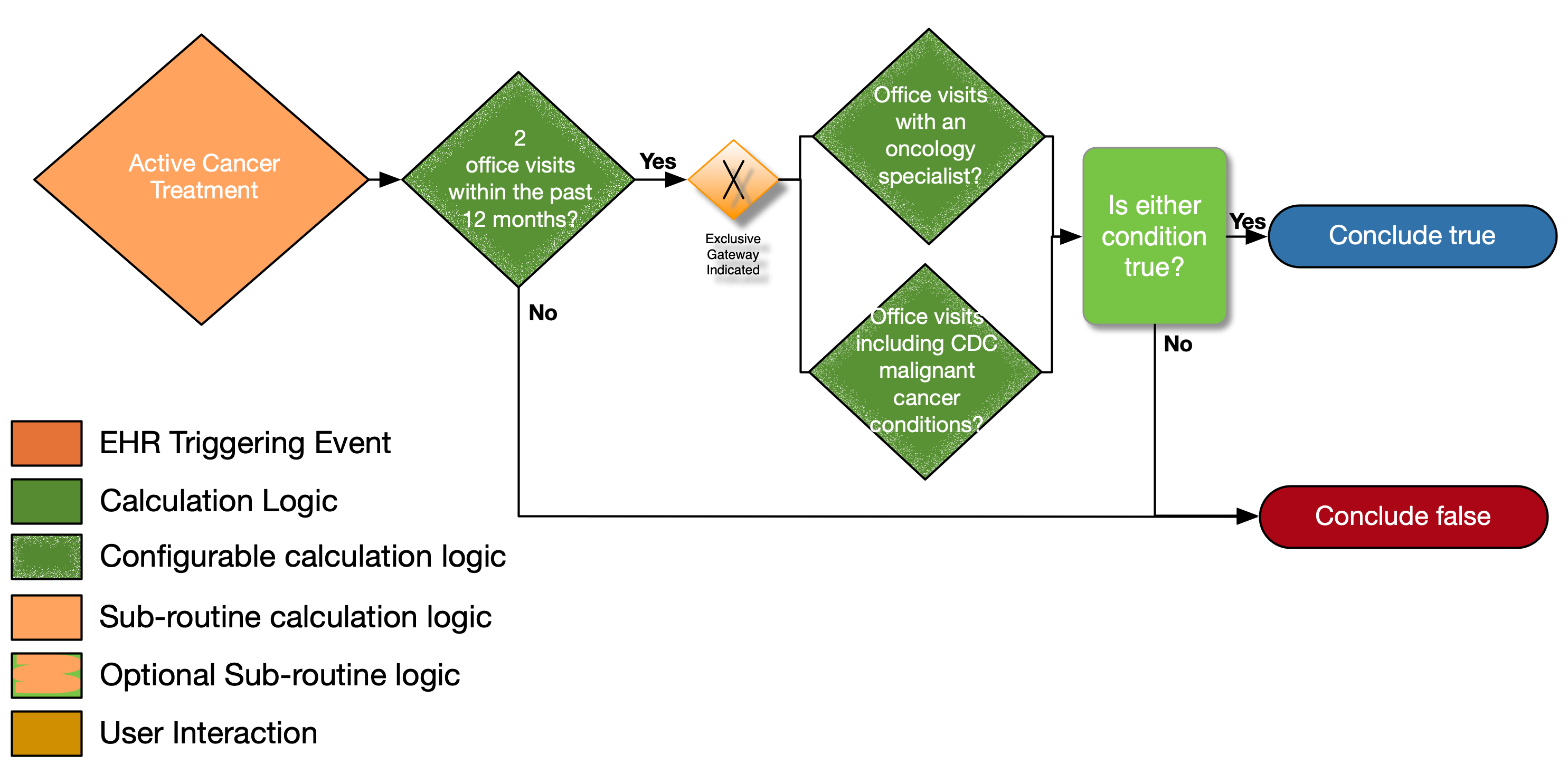
| Definition | Answer to Proceed | Details | Data (Terminology) Requirement |
|---|---|---|---|
| Two office visits within the past 12 months? | No | Look for a minimum of two distinct encounters within 12 months of the date of the current visit for which each of the following is true: • the encounter diagnosis (primary or secondary or co-morbidity diagnosis) is listed in the CDC Malignant Cancer Conditions value set |
Office Visit |
| Office visits with an oncology specialist present? | No | • The encounter is performed by an oncologist as defined in the oncology specialty designations using the National Uniform Claim Committee (NUCC) classifications |
Oncology specialty designations (NUCC) |
| Office visits including CDC malignant cancer condition? | No | • The encounter diagnosis (primary or secondary or co-morbidity diagnosis) is listed in the CDC Malignant Cancer Conditions value set | CDC malignant cancer conditions |
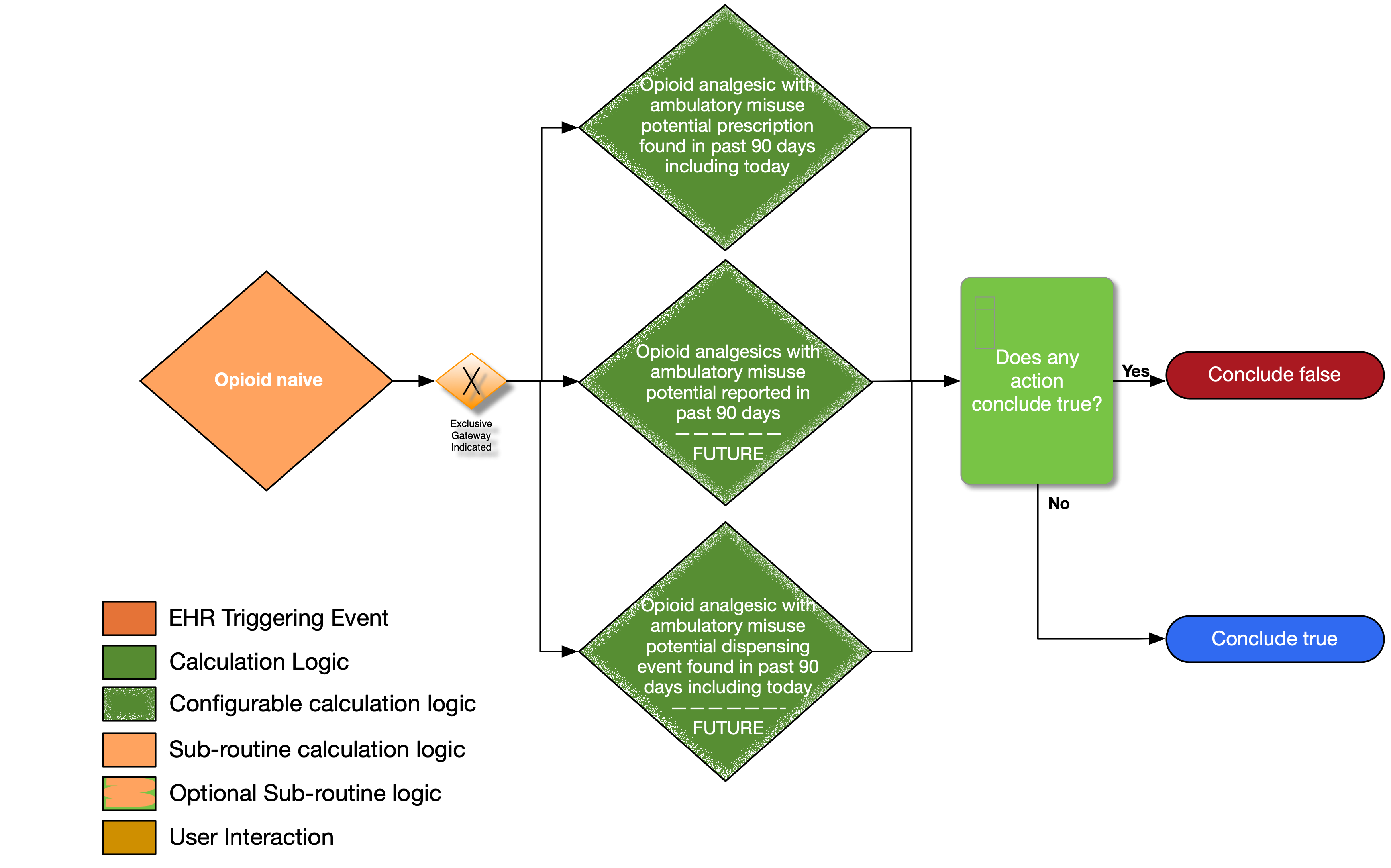
| Definition | Answer to Proceed | Details | Data (Terminology) Requirement |
|---|---|---|---|
| Opioid analgesics with ambulatory misuse potential prescription in past 90 days excluding the last 24 hours? | No | Look for an existing prescription (order) for opioid that is in the value set for opioid with ambulatory misuse potential authored within the past 90 days (excluding the last 24 hours) |
Opioid analgesics with ambulatory misuse potential |
| Opioid analgesics with ambulatory misuse potential reported as being taken excluding the last 24 hours? | No | Look for evidence of active medication on the medication list that is in the value set for opioid with ambulatory care misuse potential (excluding the last 24 hours) |
Opioid analgesics with ambulatory misuse potential |
| Opioid analgesics with ambulatory misuse potential dispensing event in past 90 days excluding the last 24 hours? | No | Look for evidence of a dispensing event for medication that is in the value set for opioid with ambulatory use potential occurring within the past 90 days (excluding the last 24 hours) |
Opioid analgesics with ambulatory misuse potential |
The following artifacts formalize the description of the logic and behavior defined by this recommendation.
| Resource | Type | Description |
|---|---|---|
| 2016 CDC Guideline Recommendation #2 | PlanDefinition | Event-Condition-Action rule that implements behavior for 2016 CDC Guideline Recommendation #2 |
| Recommendation #2 - Opioid Therapy Goals Discussion | Library | Defines the data requirements to support evaluation of recommendation #2 |
| Opioid Terminology Management Knowledge-base Data (OMTK) Library | Library | CQL Library that provides logic for implementation of opioid management functionality including Milligram Morphine Equivalents (MME). |
| Opioid Terminology Management Knowledge-base (OMTK) Library | Library | CQL Library that provides logic for implementation of opioid management functionality including Milligram Morphine Equivalents (MME). |
| Common Opioid Decision Support Logic | Library | CQL Library that provides common logic for the recommendations |
| Common OpioidCDS Configuration Logic | Library | CQL Library that provides common configuration logic for the recommendations |
| Common OpioidCDS Routines Logic | Library | CQL Library that provides common routines logic for the recommendations |
| FHIRHelpers Conversion Logic | Library | CQL Library that defines functions to convert between FHIR data types and CQL system-defined types, as well as functions to support FHIRPath implementation |
| Description | CDS Hooks Request | Expected Response |
|---|---|---|
| Patient is 18 years or older and has an outpatient Medication Request of Suboxone 2 MG / 0.5 MG Sublingual Film (RXNorm 1010603) authored "Today" as well as an Opioid Treatment Care Plan updated 89 days prior to today. Patients "Dosage Instructions" are 1 Film per day for 30 days. This patient will not trigger any cards and there should not be a recommendation message. | Request JSON example-rec-02-in-careplan-revisited | Response JSON example-rec-02-in-careplan-revisited |
| Patient is 18 years or older and has an outpatient Medication Request of Suboxone 2 MG / 0.5 MG Sublingual Film (RXNorm 1010603) authored "Today" as well as a Care Plan most recently updated 120 days prior to today. Patients "Dosage Instructions" are 1 Film per day for 30 days. Due to the most recent review of the Care Plan having been conducted more than 89 days prior to today this will result in a summary message: "Recommend creation, review or update of opioid treatment plan." | Request JSON example-rec-02-out-careplan-not-revisited | Response JSON example-rec-02-out-careplan-not-revisited |
IG © 2019+ Centers for Disease Control and Prevention (CDC). Package fhir.cdc.opioid-cds-r4#2016.4.0 based on FHIR 4.0.1. Generated 2025-09-22
Links: Table of
Contents |
License |
QA Report
| New Issue | Issues
| Version History |