Data Exchange For Quality Measures Implementation Guide, published by HL7 International / Clinical Quality Information. This guide is not an authorized publication; it is the continuous build for version 1.0.0-ballot built by the FHIR (HL7® FHIR® Standard) CI Build. This version is based on the current content of https://github.com/HL7/davinci-deqm/ and changes regularly. See the Directory of published versions
A Learning Health System (LHS) is one in which science, informatics, incentives, and culture are aligned to enable continuous learning and improvement. In an LHS, data generated from routine care, patient experiences, and health system operations are systematically collected, analyzed, and translated into knowledge that is rapidly fed back into practice. This cyclical process allows the health system to learn from every patient encounter and continuously improves health outcomes, care quality, and efficiency.
Quality measurement enables the LHS cycle by identifying variation in care, revealing gaps between current and best practices, and establishing benchmarks against which change can be evaluated.
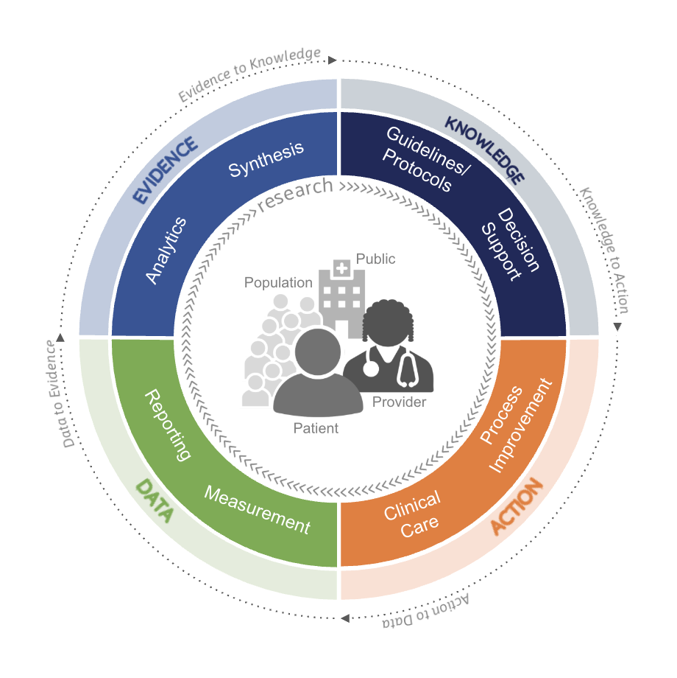
Data to Knowledge in the LHS: Within the LHS model, the data to knowledge segment represents the critical transformation of routinely collected health data into actionable insights that can improve care delivery, outcomes, and health system performance. Quality measures are foundational in this transformation providing the structured, standardized metrics needed to convert raw clinical, operational, and patient-generated data into meaningful indicators of performance, safety, effectiveness, equity, and value.
Quality measures create feedback loops between the various segments of the LHS: insights derived from measurement inform practice changes, policy decisions, and system redesign, while subsequent data collection assesses the impact of those changes. In this way, quality measurement acts as both the lens and the engine of learning—ensuring that the progression from data to knowledge is systematic, transparent, and continuously aligned with the goals of better health outcomes, improved patient experience, and more efficient care delivery.
Knowledge to Action in the LHS: In the knowledge to action segment of the LHS, evidence and insights generated through analysis of measure reports are operationalized to change clinical practice. Quality measures play a pivotal role in this phase by serving as the bridge between clinical knowledge (such as evidence‑based guidelines) and actionable, point‑of‑care interventions delivered through Clinical Decision Support (CDS) tools. Quality measures distill complex clinical guidelines into specific, observable, and measurable care processes or outcomes. By defining what “high‑quality care” looks like in operational terms (e.g., appropriate medication use, timely screenings, or adherence to care pathways), quality measures provide the structured logic needed for CDS systems to act on guidelines. This translation ensures that abstract recommendations are converted into concrete clinical targets that can be monitored and improved.
Action to Data in the LHS: This component of the LHS is where changes in clinical practice resulting from effective quality measurement generate new data that feed subsequent learning cycles. Quality measures play a critical enabling role by shaping what data are collected, how consistently those data are captured, and whether the resulting data are fit for purpose.
Measure specifications translate recommended clinical actions into explicit documentation requirements—such as structured fields for laboratory results, medications, diagnoses, timing, and exception handling—thereby informing EHR configuration, clinical template and workflow design. The standardized data definitions, inclusion and exclusion criteria, and use of terminology embedded in measures promote consistent and interpretable documentation across all care settings, improving data accuracy, comparability, and reliability.
Finally, because performance reporting and audit processes are typically built upon precise and unambiguous measure specifications, they enable continuous validation loops that surface missing data, inconsistent documentation, and anomalous trends. These insights support targeted workflow adjustments and training ensuring that each cycle produces more complete, timely, and trustworthy data. By strengthening both data availability and data quality, measure specifications ensure that care delivery meaningfully contributes to continuous learning and quality improvement.
Creating Feedback Loops for Continuous Learning: Quality measures enable closed‑loop learning by providing feedback on whether CDS‑driven actions lead to measurable improvement. Performance data can be fed back to clinicians, teams, and system leaders through dashboards or reports, reinforcing desired behaviors and highlighting areas needing refinement. These feedback loops allow CDS logic, clinical workflows, and even guideline interpretations to be continuously updated based on real‑world outcomes.
Quality measures are essential enablers of a Learning Health System. They translate evidence based guidelines into computable, actionable elements that power CDS interventions, guide clinician behavior at the point of care, and generate feedback on impact. Through this role, quality measures help ensure that knowledge is not only generated, but reliably and efficiently transformed into improved clinical practice and patient outcomes.
This implementation guide is part of a larger FHIR-based quality improvement and quality measurement standards landscape, depicted in the following diagram:

The left side of the quality measurement standards landscape diagram depicts the activities and standards associated with measure specification, while the right side depicts measure reporting. Stakeholders and the roles they play are represented by the three rounded rectangles in the foreground. Note that the lists are representative of typical stakeholders, but that a single stakeholder may play any or all of the roles in this diagram. For example, an institution specifying its own measures for internal use would be the Producer, Consumer, and Specifier.
Measure specification involves the end product of the measure development process, a precisely specified, valid, reliable, and clinically significant measure specification to support accurate data representation and capture of quality measures. Clinical quality measures (CQMs) are tools that help measure and track the quality of health care services provided in care delivery environments, including eligible clinicians (ECs), eligible hospitals (EHs), and critical access hospitals (CAHs). Measuring and reporting CQMs helps to ensure that our health care system is delivering effective, safe, efficient, patient-centered, equitable, and timely care. CQMs measure many aspects of patient care, including patient and family engagement, patient safety, care coordination, public health, population health management, efficient use of healthcare resources, and clinical process and effectiveness. For more information on the basics of Clinical Quality Measures, see Clinical Quality Measures Basics. Before Electronic Health Record (EHR) systems, chart-abstracted CQMs were predominant. Modern EHR systems enable electronic CQMs, or eCQMs.
Measure reporting involves the data collection and aggregation, calculation and analytics, and ultimately reporting of quality measures. Measure reporting may be accomplished in different ways at various levels of the healthcare delivery system, from individual providers attesting to specific quality measures as part of federally-regulated healthcare quality initiatives, to provider organizations reporting to healthcare plans as part of payer quality improvement activities, to institutions reporting on the quality of their own healthcare delivery.
Stakeholders in the quality space, represented by the three rounded rectangles in the foreground of the above diagram, fall into three broad categories:
Data Producers in the diagram represent the various stakeholders involved in the de novo creation of healthcare data. Data Producers can include providers and provider systems; patients, care teams, caregivers, and patient engagement systems; and other related clinical systems such as laboratory, clinic, and hospital information systems that are primary producers of patient healthcare information.
Data Consumers in the diagram represent the various stakeholders involved in the consumption and use of healthcare data. Data Consumers can include data routers and aggregators, payers, health information exchanges and health integrated networks, as well as public health and other healthcare-related agencies.
Specifiers in the diagram represents the various stakeholders involved in the specification of quality measures for use in healthcare quality measurement and reporting. Specifiers can include quality agencies, public health, and other healthcare-related agencies, industry consortiums concerned with improving care quality, and clinical professional societies. Specifiers may also be institutions and clinics using the quality measurement standards to specify quality measures for use in their own environments and quality improvement initiatives.
The shaded areas underlying the stakeholders depict the various standards involved (see Clinical Quality Framework for more information).
Fast Healthcare Interoperability Resources, or FHIR, is a Health Level 7 (HL7) platform specification for healthcare that supports exchange of healthcare information between systems. FHIR is universally applicable, meaning that it can be used in a broad variety of implementation environments. The platform provides layers of implementation that support foundational protocols; base implementation functionality such as conformance and terminology; administrative functionality to represent patients, care teams, locations, and organizations; healthcare processes including clinical and diagnostic information, as well as medication, workflow, and financial; and finally, a clinical reasoning layer that provides support for the representation of knowledge and reasoning about healthcare.
The quality measurement standards landscape makes use of all these layers of FHIR: the foundational and implementation layers to define interactions and profiles; the administrative and process layers to represent the data of interest for quality measurement; and the clinical reasoning layer to specify and support evaluation and reporting of quality measures.
Clinical Quality Language, or CQL, is an HL7 cross-paradigm specification that defines a high-level, domain-specific language focused on clinical quality and targeted for use by measure and decision support artifact authors. In addition, the specification describes a machine-readable canonical representation called Expression Logical Model (ELM) targeted at implementations and designed to facilitate sharing and evaluation of clinical knowledge.
This ability to render clinical knowledge in a high-level human-readable form as well as an intermediate-level, platform-independent machine-readable form makes CQL an ideal mechanism for specifying the criteria involved in quality measures.
The FHIR Quality Measure Implementation Guide defines conformance profiles and guidance focused on the specification of quality measures using the FHIR Measure and Library resources. The IG does not standardize the content of any particular measure, rather it defines the standard approach to the representation of that content so that quality measure specifiers can define and share standardized FHIR-based electronic Clinical Quality Measures (eCQMs).
The Quality Improvement Core Implementation Guide, or QI-Core, defines a set of FHIR profiles with extensions and bindings needed to create interoperable, quality-focused applications. Importantly, the scope of QI-Core includes both quality measurement and decision support to ensure that knowledge expressed can be shared across both domains. QI-Core is derived from US Core, meaning that where possible, QI-Core profiles are based on US Core to ensure alignment with and support for quality improvement data within healthcare systems in the US Realm.
The Data Exchange for Quality Measures Implementation Guide, or DEQM, (this IG) provides a framework that defines conformance profiles and guidance to enable the exchange of quality information and quality measure reporting (e.g. for transferring quality information from a health care provider to a payer). The DEQM expects to use quality measures specified in accordance with the Quality Measure IG and QI-Core.
The quality improvement ecosystem covers every aspect of the healthcare delivery system, and needs to be able to represent information across that entire spectrum. FHIR provides a foundation for representation of this information in a universally applicable way. In particular cases, more specificity is required to capture the intended meaning of healthcare information. As FHIR is more and more broadly adopted, consensus among participating stakeholders on the use of particular profiles and patterns enables semantic interoperability for more use cases.
Within the US Realm, US Core profiles comprise this base consensus, and although it enables a variety of interoperability use cases, the profiles do not represent all of the requirements for quality improvement. The QI-Core profiles are derived from US Core and provide this additional functionality.
There are occasional instances where additional specificity or functionality is required explicitly for quality measurement, or a particular component within a quality measure. In these cases, additional profiles are defined within the DEQM, or by stakeholders such as measure developers or implementers. For example, the Medication Reconciliation Post Discharge measure example included in this implementation guide references the Healthcare Effectiveness Data and Information Set (HEDIS) Implementation Guide, which defines profiles specific to that particular HEDIS measure.
The following diagram depicts this data model standards landscape:
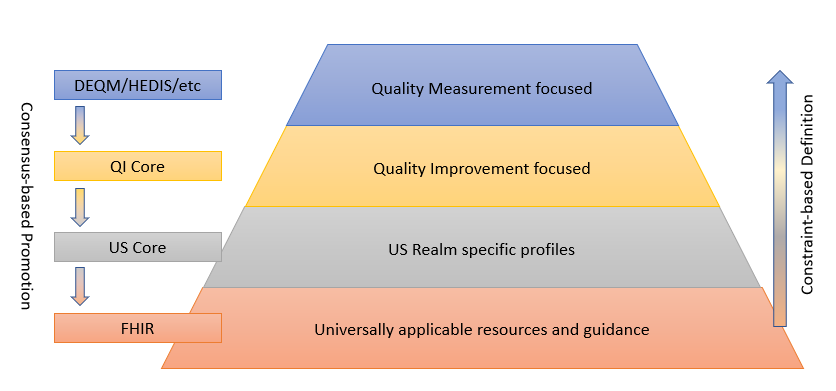
As illustrated, FHIR provides the foundation, and sets of profiles are built on top of FHIR that provide more and more focused use cases by constraining profiles and extending functionality to cover gaps. While the additional layers are necessary to represent specific operations and provide space for agreement among relevant stakeholders, the consensus-based standards development process is used to suggest changes to the layers below, resulting in an ever-broadening umbrella of interoperability.
This layering of profiles balances the relative adoption and implementation maturity of FHIR and the data representation requirements of the use cases involved, guided by the following principles: