HL7 Europe Laboratory Report, published by HL7 Europe. This guide is not an authorized publication; it is the continuous build for version 2.0.0 built by the FHIR (HL7® FHIR® Standard) CI Build. This version is based on the current content of https://github.com/hl7-eu/laboratory/ and changes regularly. See the Directory of published versions
Logical data models (or information models) for the European Health Data Space (EHDS) are currently being developed by the Xt-EHR Joint Action. These models will form the basis of the Joint Action proposal for the future EHDS Implementing Act.
They represent evolving, refined interpretations of the data sets described in the eHealth Network (eHN) Guidelines, and are expected to be further updated during the Joint Action process.
This Implementation Guide (IG) aims to align with the emerging EHDS logical models and to provide HL7 FHIR profiles that realise the requirements identified in these models.
Below is a simplified representation of a laboratory report. The data set comprises several basic parts, as visualised in the diagrams below.
Figure 1: Laboratory dataset model
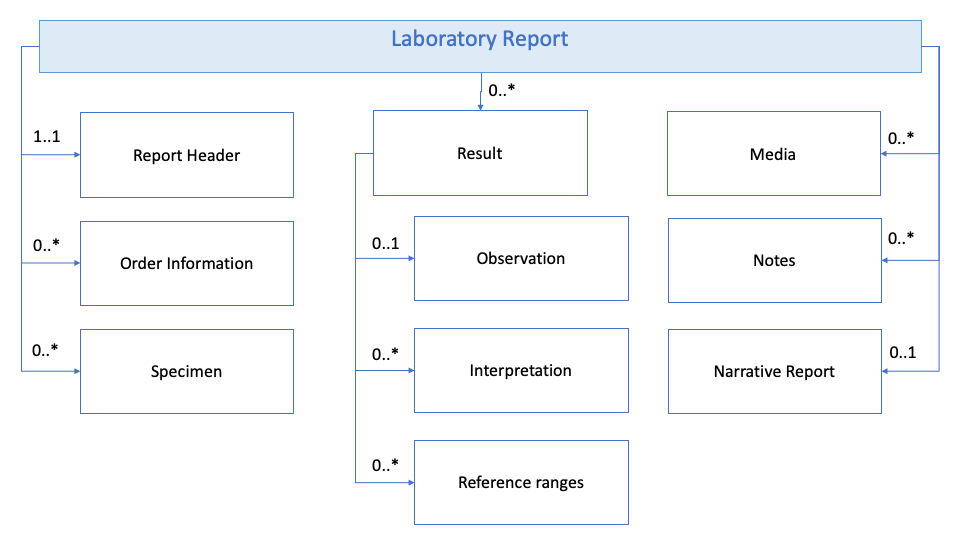
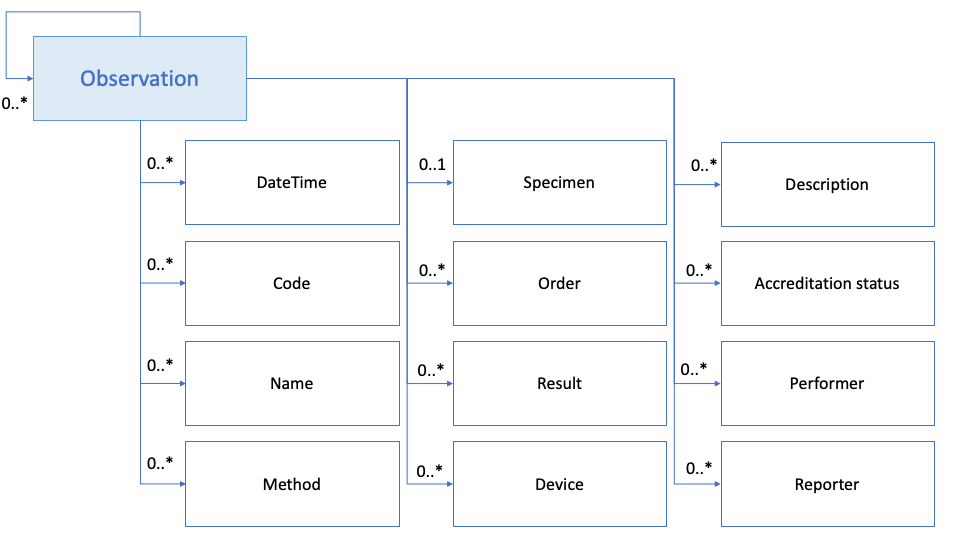
Figure 2: Laboratory observation dataset model
The EHDS logical models currently supported in this version of the guide are listed below. For the most recent and in-progress versions, please refer to the Xt-EHR EHDS Logical Information Models Implementation Guide.
| Model | Description |
|---|---|
| EHDSLaboratoryReport | EHDS logical model for laboratory result reports |
| EHDSLaboratoryObservation | EHDS refined base model for observations performed by the laboratory |
| Model | Description |
|---|---|
| EHDSPatient | EHDS refined base model for Patient (subject of care) |
| EHDSRelatedPerson | EHDS refined base model for a person related to the patient |
| EHDSHealthProfessional | EHDS refined base model for Health Professional |
| EHDSOrganisation | EHDS refined base model for healthcare organisations and providers |
| EHDSAttachment | EHDS refined base model for containing or referencing attachments. |
| EHDSDevice | EHDS refined base model for Device information |
| EHDSCondition | EHDS refined base model for a clinical condition, problem, diagnosis, or other event, situation, issue, or clinical concept that has risen to a level of concern |
| EHDSMedicationAdministration | EHDS refined base model for medication administered to the patient |
| EHDSImmunisation | EHDS refined base model for immunisation |
| EHDSObservation | EHDS refined base model for medical test results and other clinical observations |
| EHDSSpecimen | EHDS refined base model for specimen information |
| EHDSServiceRequest | EHDS refined base model for requested laboratory services |