Response to Regulatory Questions (RTQ), published by HL7 International. This guide is not an authorized publication; it is the continuous build for version 0.1.0 built by the FHIR (HL7® FHIR® Standard) CI Build. This version is based on the current content of https://github.com/HL7/rtq-ig/ and changes regularly. See the Directory of published versions
Home
| Official URL: https://build.fhir.org/ig/HL7/rtq-ig/ImplementationGuide/hl7.fhir.uv.rtq-ig | Version: 0.1.0 | |||
| Draft as of 2026-03-24 | Computable Name: RTQ_IG | |||
Introduction
The Response to Question (RTQ) Implementation Guide is the standardized framework for structuring regulatory questions and responses to regulatory questions.
Built on HL7 FHIR R5, it replaces static Word and PDF question/response documents with traceable, machine-readable FHIR resources, enabling precise tracking, automated triage, and global harmonization.
RTQ encapsulates the entire question (e.g., cover letter, questions, supporting attachments, and metadata) and the response to questions (e.g., cover letter, response to questions, supporting attachments, and metadata).
This guide defines the technical framework to create a seamless Q&A loop between Regulators and Applicants.
✨ See It In Action
Experience the power of structured FHIR data rendered into high-fidelity, interactive dashboards.
Why Adopt RTQ?
Structured & Traceable. Every question is a discrete Questionnaire item; every answer is a linked QuestionnaireResponse. No more lost questions or ambiguous email threads.
Harmonized Across Agencies. One standard format for all international regulators. Build your system once to handle questions from all regulators.
Analytics-Ready. Because questions are structured data, instantly visualize trends: response cycle times, most frequent question topics, regional variances, and predict questions in advance or response quality.
Automated Workflow. Route questions to the correct SME based on metadata (e.g., each question is tagged with a specific CTD section), reducing triage time from days to minutes.
Background
In biopharmaceutical regulatory affairs, health authorities routinely issue formal questions during the review of regulatory applications. Traditionally, these are exchanged as unstructured Word or PDF documents via email or portals. This results in high manual effort, limited traceability, and "dead data" that cannot be analyzed.
RTQ changes that by modeling questions as Questionnaires and responses as versioned QuestionnaireResponses, leveraging the FHIR standard to align regulatory Q&A processes with the modern digital systems.
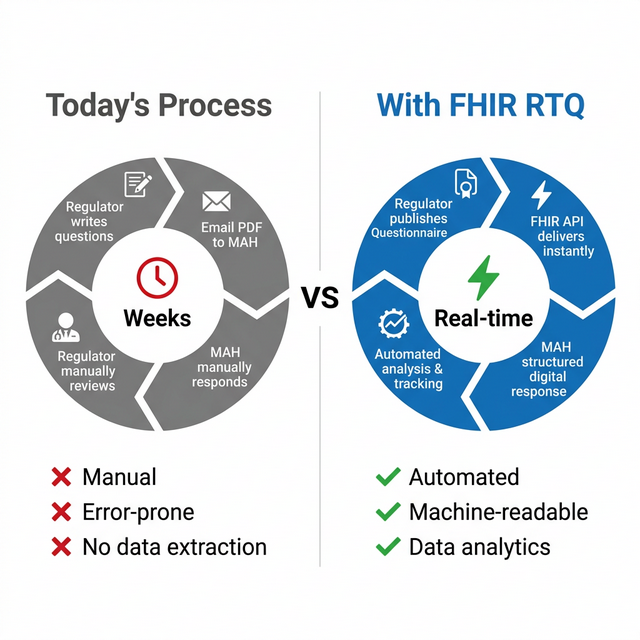
Figure 1: Manual PDF exchange vs. FHIR RTQ — from weeks to real-time
This aligns with the broader strategy defined in the APIX Implementation Guide: moving towards the "Real-time algorithmic exchange" envisioned in the 2023 International Journal of Pharmaceutics framework (click to read).
Just as ISO 20022 harmonized global payments, RTQ + APIX harmonizes the regulatory dialogue—standardizing the content (RTQ) and the exchange (APIX) to unlock sub-second workflows.
| ✅ In Scope | ❌ Out of Scope |
|---|---|
|
|
Governance & Collaboration
RTQ is developed under the HL7's Biomedical Research and Regulation (BR&R) Working Group with active participation from regulators, pharmaceutical companies, and technology vendors.
All meetings are public; notes and recordings are available via HL7 BR&R Working Group's RTQ project page.
Get Involved
- Join the weekly calls.
- Test the reference implementation.
We welcome industry, solution providers, and regulators from every region to contribute to this global standard.