Making Electronic Data More available for Research and Public Health (MedMorph), published by HL7 International - Public Health Work Group. This is not an authorized publication; it is the continuous build for version 1.0.0). This version is based on the current content of https://github.com/HL7/fhir-medmorph/ and changes regularly. See the Directory of published versions
| Official URL: http://hl7.org/fhir/us/medmorph/ImplementationGuide/hl7.fhir.us.medmorph | Version: 1.0.0 | |||
| Active as of 2023-06-08 | Computable Name: MakingElectronicDataMoreAvailableForResearchAndPublicHealth | |||
The Making Electronic Data More Available for Research and Public Health (MedMorph) project seeks to use emerging health data and exchange standards, namely Health Level 7 (HL7®) Fast Healthcare Interoperability Resources (FHIR®), to develop and implement an interoperable solution to enable access to clinical data. The MedMorph project fits within the Centers for Disease Control and Prevention (CDC) strategic imperative of transforming how data are collected, used, and shared through modern Information Technology (IT) capabilities to save lives and improve health. The initial phase of the MedMorph project was funded by the Health and Human Services (HHS) Assistant Secretary for Planning and Evaluation (ASPE) Patient-Centered Outcomes Research Trust Fund (PCORTF) and executed by CDC’s Center for Surveillance, Epidemiology, and Laboratory Services (CSELS) Public Health Informatics Office (PHIO) to advance research and public health goals.
The project aims to leverage the maturation of standards and requirements for certification of health information technology by the Office of the National Coordinator for Health IT (ONC) that underpin many EHRs. ONC’s 2015 Edition Health IT Certification Criteria and the Cures Update include, but are not limited to, the United States Core Data for Interoperability (USCDI) Version 1 and the use of standardized application programming interfaces (API) for patient services (such as FHIR R4 and Bulk FHIR APIs). Broader implementation of these certification criteria has created a health IT environment ripe for developing standards-based scalable and extensible solutions to overcome interoperability challenges.
The MedMorph project modeled four use cases that are representative of broader areas of research and public health: Chronic Hepatitis C (Hep C) Surveillance, an infectious disease; Central Cancer Registry Reporting, a chronic disease; Health Care Surveys, pertaining to health care utilization and not a specific condition; and Research, supporting standardized data exchange for public health research. These representative use cases informed the design of a scalable and extensible reference architecture to facilitate interoperable data exchange for a variety of research and public health use cases. For more detailed explanation on the reference architecture refer to next paragraph and the Implementation Guide Overview section.
The Implementation Guide (IG) presented here is also known as the MedMorph Reference Architecture (RA) IG. “Reference Architecture” refers to a common framework (e.g., actors, systems, FHIR resources, FHIR APIs, FHIR operations, security mechanisms) leveraged by multiple use cases. The MedMorph RA IG is accompanied by content IGs, which provide content profiles pertaining to a specific use case. Requirements for specific use cases are driven by the content IGs whereas the requirements for the framework components, such as the Health Data Exchange App (HDEA) (MedMorph’s backend services app), Knowledge Artifact Repository (KAR), Trusted Third Party (TTP), etc., are identified as actors in the MedMorph RA. There are example capability statements created for actors whose specific requirements would be detailed in the content IGs.
NOTE: Implementers of the MedMorph RA IG have to use one or more content IGs to operationalize the framework as this IG provides only the framework and does not provide all the necessary content profiles that will be required to implement a use case.
Examples of Content IGs include Central Cancer Registry Reporting IG and Health Care Survey Reporting IG and Research Data Exchange IG. The relationship between these IGs is further explained in the IG Types and their Relationships section section. An overview of this MedMorph RA IG actors, systems and interactions are explained further in the MedMorph Actors and Systems section and a thumbnail is provided below for reference.
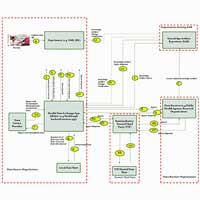
This IG depends on FHIR R4 (4.0.1), US Core 3.1.1, Bulk Data IG 1.0.1, SMART App Launch IG 2.0.0 and Subscriptions Backport IG 1.1.0. The reason for depending on the above specific versions of the specifications and implementation guides is to be compatible with the US regulatory requirements related to USCDI and Cures Update.
The main sections of this IG are: