

This fragment is available on en/profiles.html
This publication includes IP covered under the following statements.
| Type | Reference | Content |
|---|---|---|
| web | browser.ihtsdotools.org | http://snomed.info/sct |
| web | www.lilly.com | https://www.lilly.com |
| web | github.com | Clinical Study Schedule of Activities, published by HL7 International / Biomedical Research and Regulation. This guide is not an authorized publication; it is the continuous build for version 2.0.0-ballot built by the FHIR (HL7® FHIR® Standard) CI Build. This version is based on the current content of https://github.com/HL7/Vulcan-schedule-ig/ and changes regularly. See the Directory of published versions |
| web | doi.org | Richardson, A., (2024) “Representing Clinical Study Schedule of Activities as FHIR Resources: Required Characteristic Attributes”, Journal of the Society for Clinical Data Management 4(2) ↩ |
| web | www.cdisc.org | The CDISC ODM is a model designed for the transportation and storage of Study Data for Clinical Studies. The landing page for the standard can be found here: https://www.cdisc.org/standards/data-exchange/odm |
| web | cdisc.org | Note: the CDISC ODM is owned by CDISC . A free CDISC website account is required to access the material - create an account here . |
| web | www.cdisc.org | Note: the CDISC ODM is owned by CDISC . A free CDISC website account is required to access the material - create an account here . |
| web | github.com | The CDISC repository for the ODM found here . |
| web | github.com | ODM Schema |
| web | www.sciencedirect.com |
For an example of a master protocol
, the existing ResearchStudy.partOf
predicate can be used for the sub-studies as part of master protocol, this allows for differentiation of eligibility criteria, lifecycle, etc.
|
| web | medinform.jmir.org | We have chosen to use the extensions proposed by Richardson A, Genyn P Clinical Trial Schedule of Activities Specification Using Fast Healthcare Interoperability Resources Definitional Resources: Mixed Methods Study JMIR Med Inform 2025;13:e71430 - henceforth referred to as MMS. The authors have kindly agreed for their work to be utilised as part of the IG, with the qualification that full recognition for the work shall remain theirs alone. |
| web | medinform.jmir.org | For a deeper discussion and definition of this, please refer to the source paper here: Clinical Trial Schedule of Activities Specification Using Fast Healthcare Interoperability Resources Definitional Resources: Mixed Methods Study . |
| web | www.cdisc.org | Using the extensions we can represent the SoA as a Graph - this is key to being able to provide decision support in a prospective manner for a user utilising a healthcare system to manage the execution of a clinical trial. There are analogues to the implementation of Timelines in the CDISC Unified Study Design Model (USDM) ; so transformations between representations of Study Designs should be mappable between the standards. |
| web | en.wikipedia.org | This drive for efficiency has given rise to adaptive designs and master protocols, such as platform , basket , and umbrella trials. These modern approaches often incorporate conditional logic, where the study path for a participant can change based on interim results, biomarker status, or other criteria. Consequently, the schedule of activities is no longer a static table but a dynamic plan with branching pathways and conditional events. While these designs can accelerate drug development and reduce overall costs, they introduce significant complexity in defining, implementing, and managing the schedule of activities across different systems. Additionally, under the guise of Adaptive Trial Design the implementation of the study can change significantly for a patient. |
| web | bmcmedicine.biomedcentral.com | This drive for efficiency has given rise to adaptive designs and master protocols, such as platform , basket , and umbrella trials. These modern approaches often incorporate conditional logic, where the study path for a participant can change based on interim results, biomarker status, or other criteria. Consequently, the schedule of activities is no longer a static table but a dynamic plan with branching pathways and conditional events. While these designs can accelerate drug development and reduce overall costs, they introduce significant complexity in defining, implementing, and managing the schedule of activities across different systems. Additionally, under the guise of Adaptive Trial Design the implementation of the study can change significantly for a patient. |
| web | database.ich.org | https://database.ich.org/sites/default/files/E6_R2_Addendum.pdf ↩ |
| web | database.ich.org | CLINICAL ELECTRONIC STRUCTURED HARMONISED PROTOCOL - M11 ↩ |
../assets/images/001.svg 
|
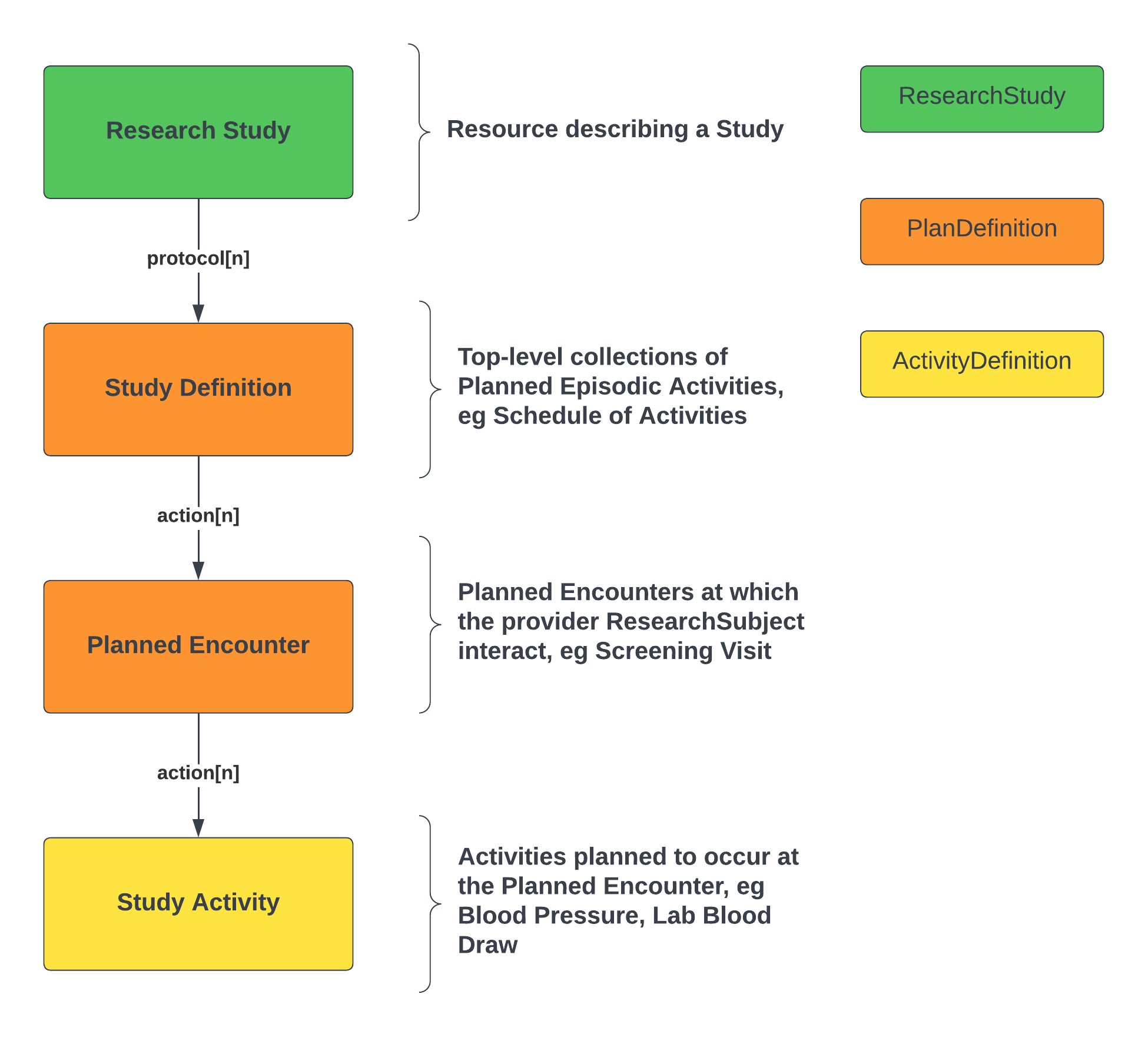
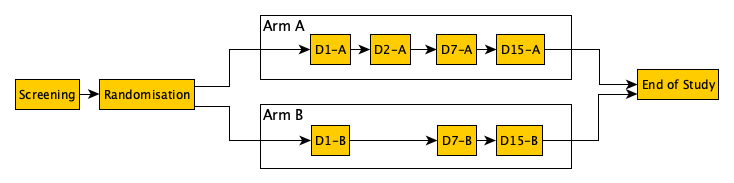
Vulcan-SoA-basic-structure.png 
|
conditional-activities.jpeg 
|
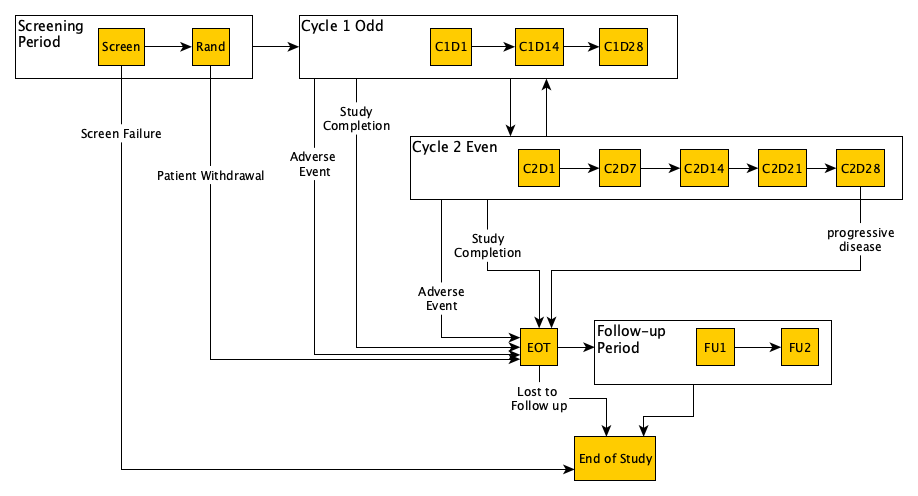
dynamic-schedules-cycles-example.png 
|
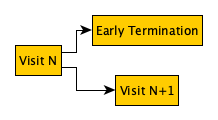
dynamic-visit-schedule-exit-example.png 
|
dynamic-visit-schedule-multiple-paths-example.png 
|
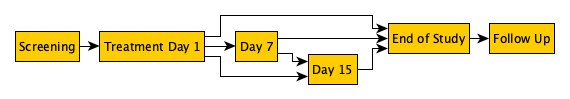
dynamic-visit-schedule-simple-example.png 
|
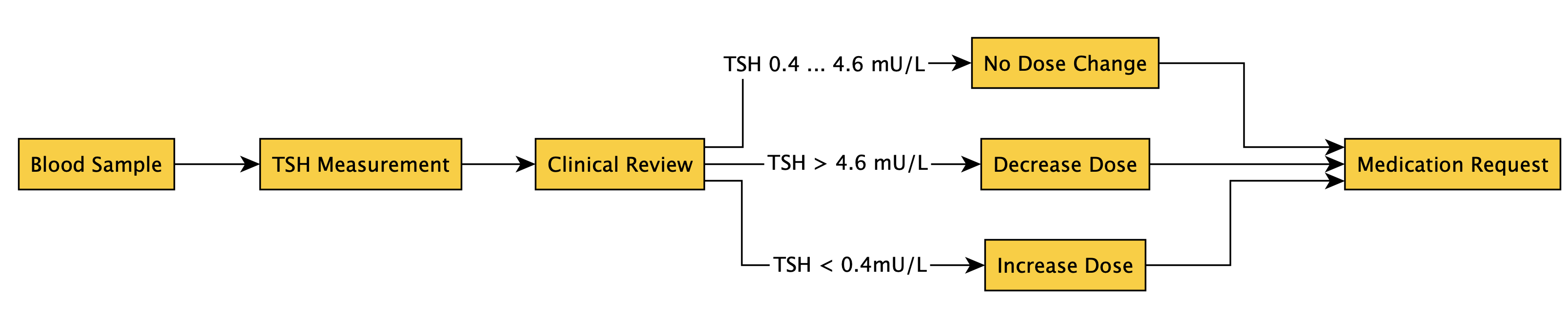
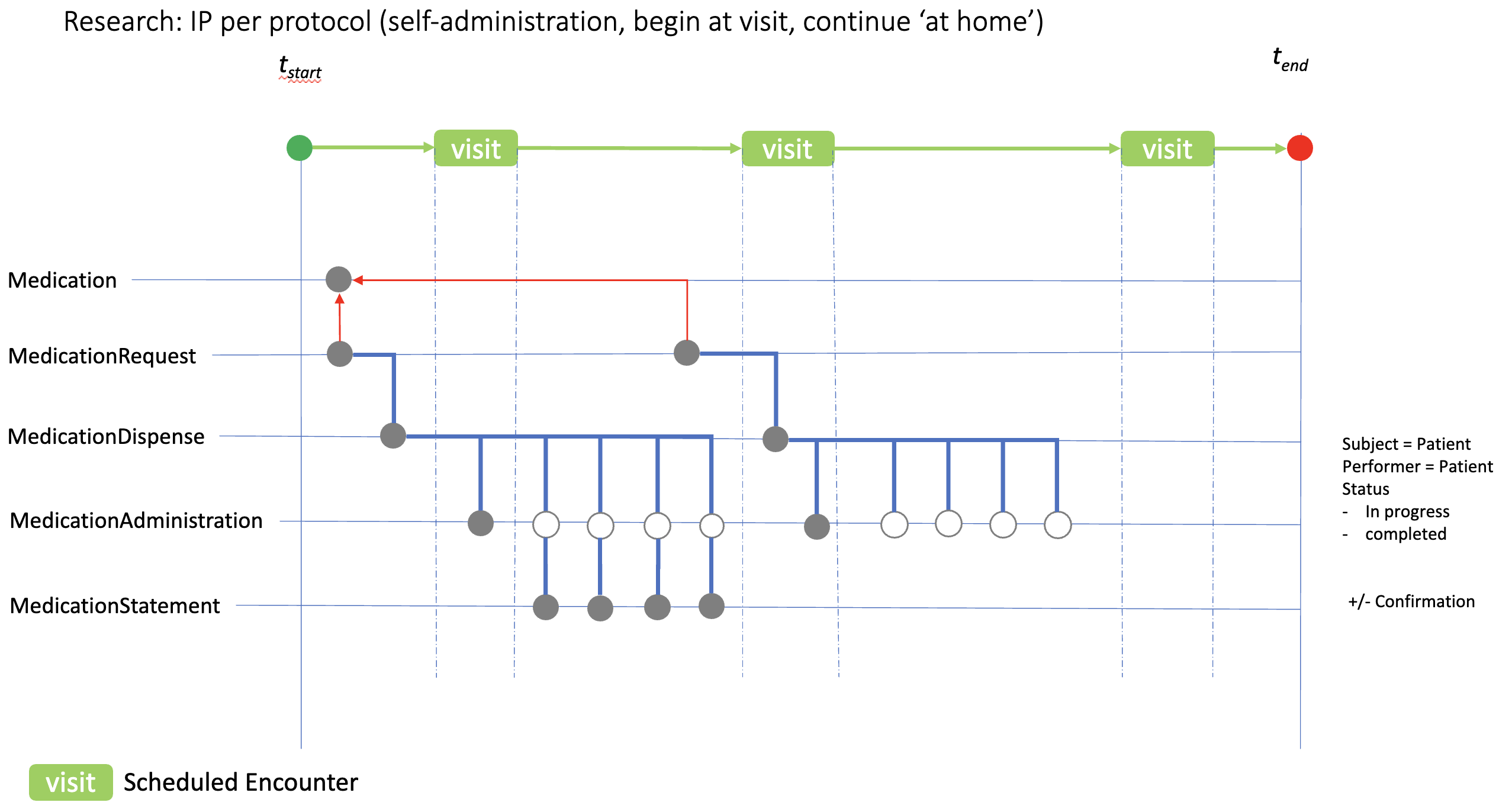
ip-admin-home-dose.png 
|
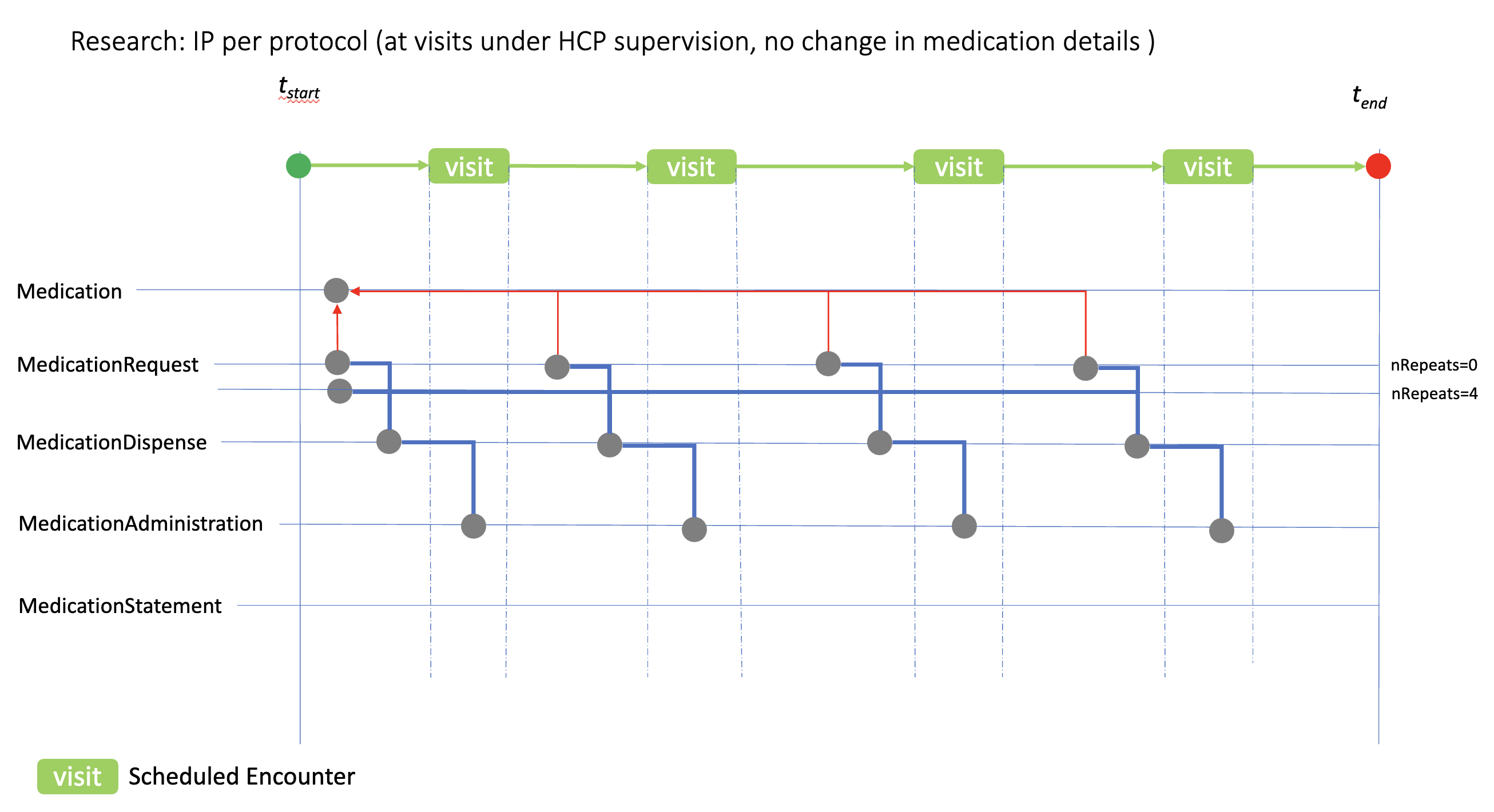
ip-admin-no-change.png 
|
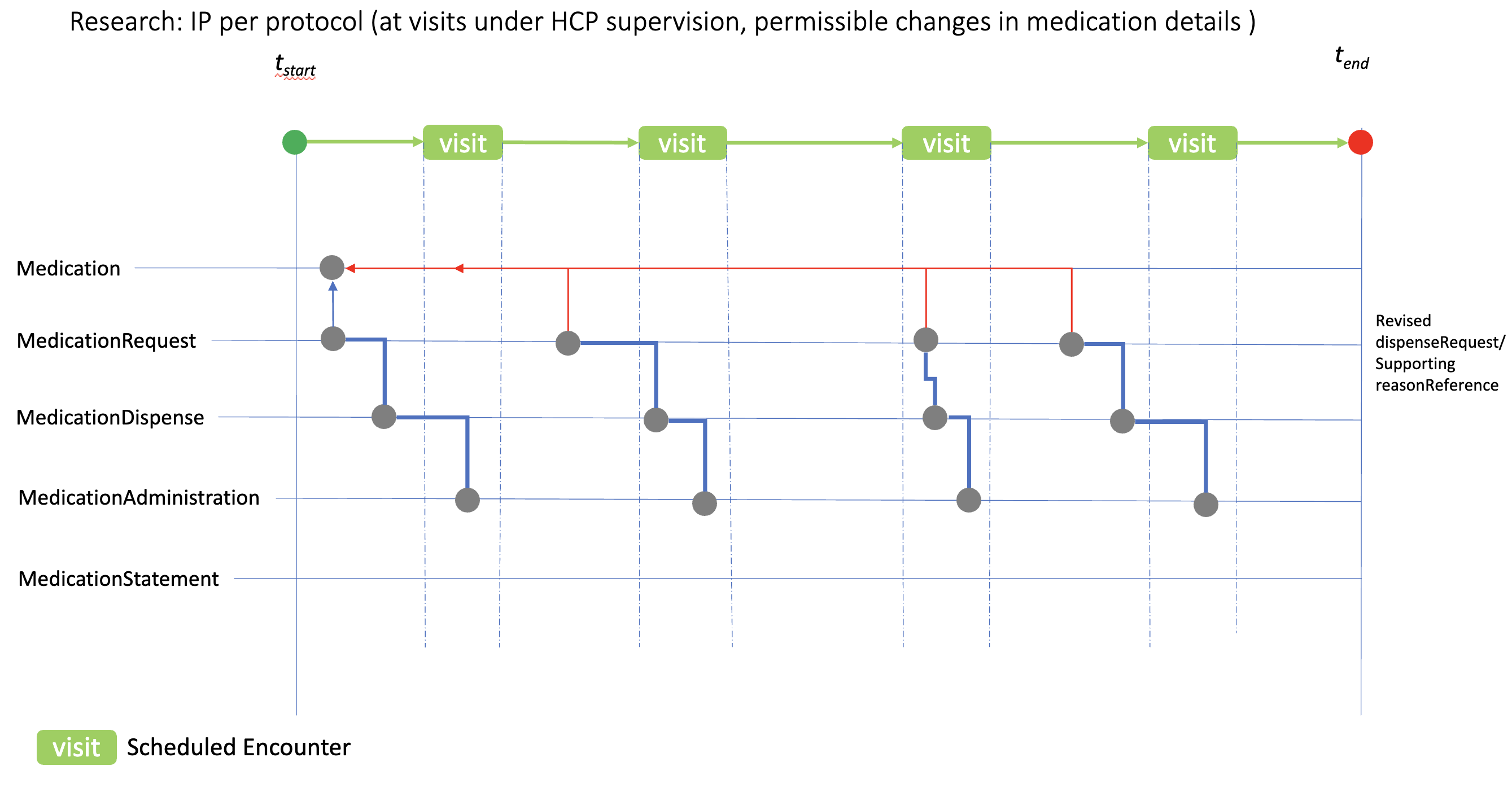
ip-admin-permissible-change.png 
|
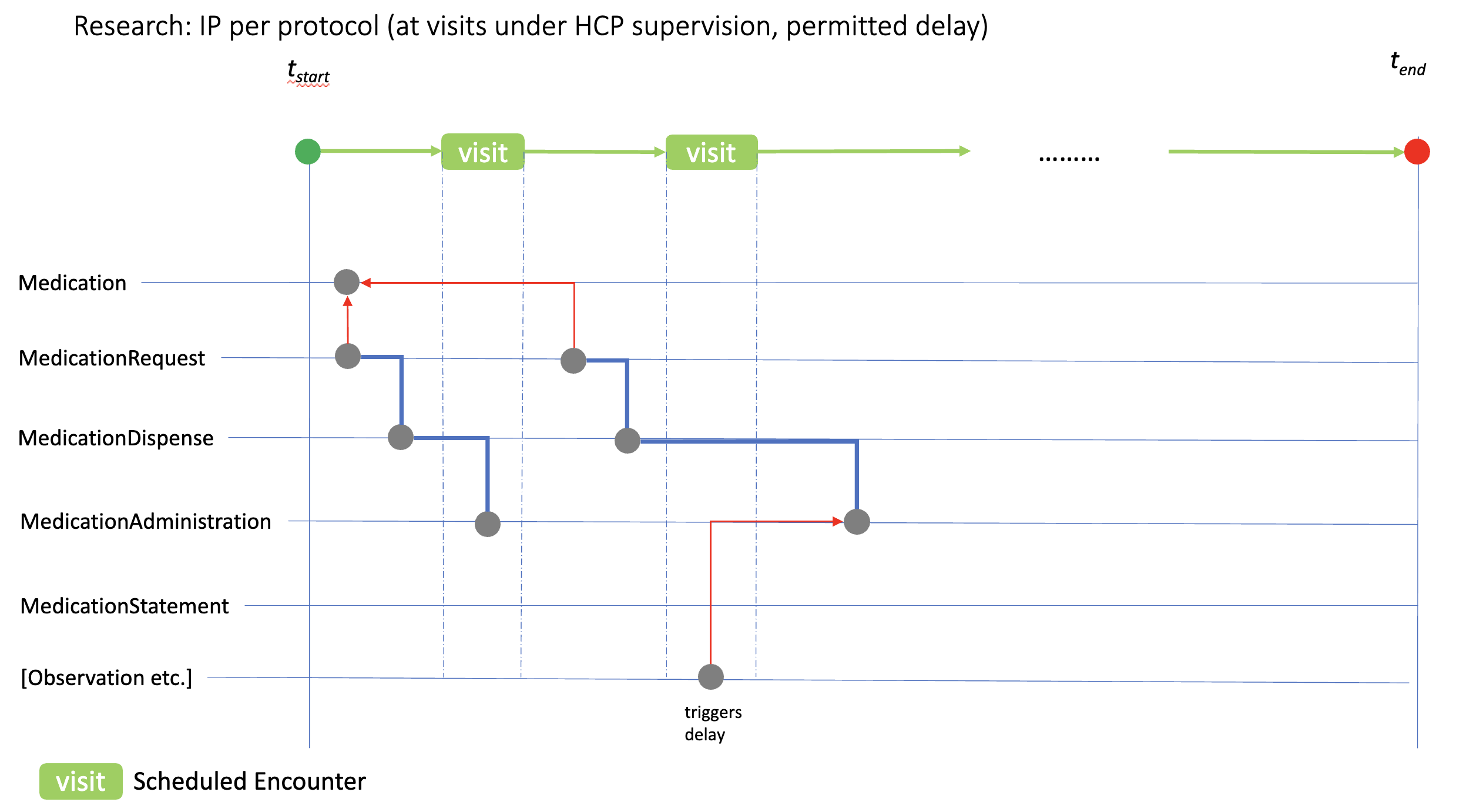
ip-admin-permissible-obs.png 
|
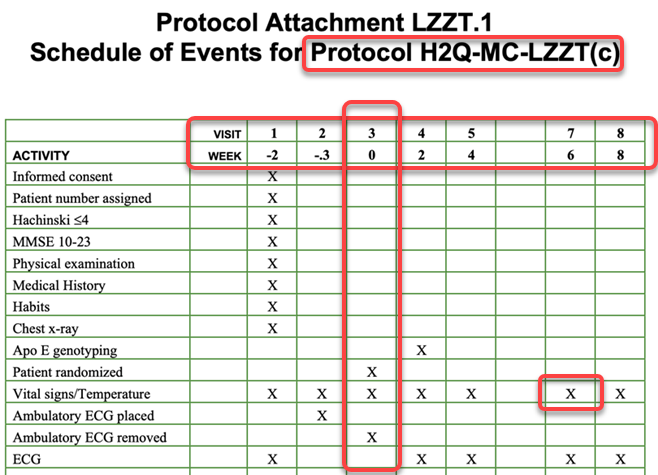
lzzt_soa_650x475_imagemap.png 
|
tree-filter.png 
|
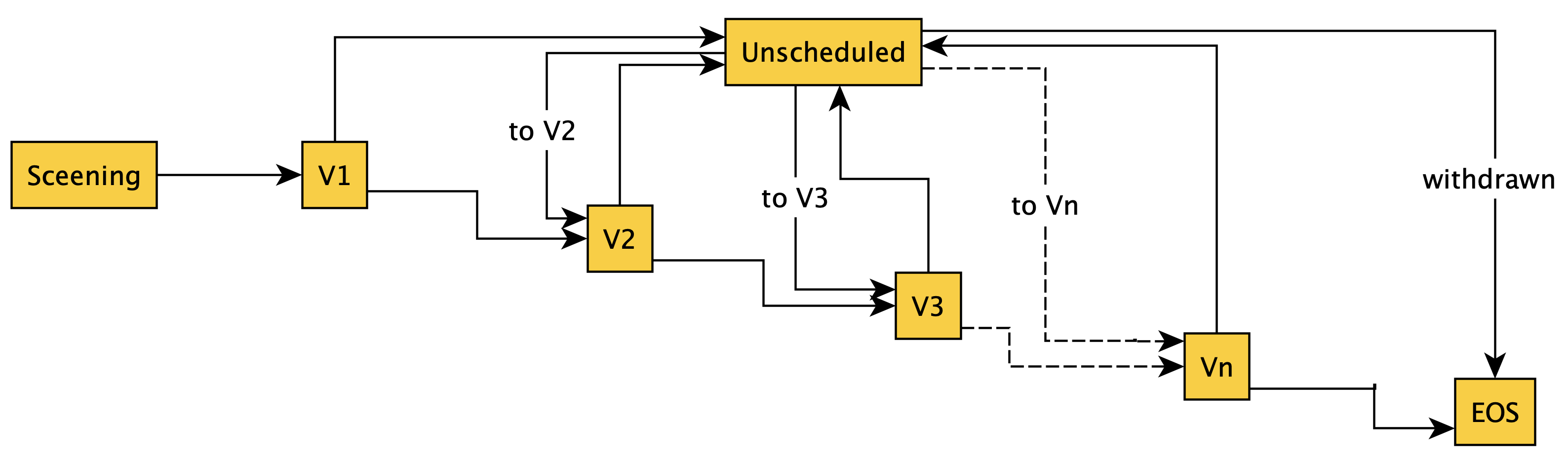
unscheduled-visits.jpeg 
|
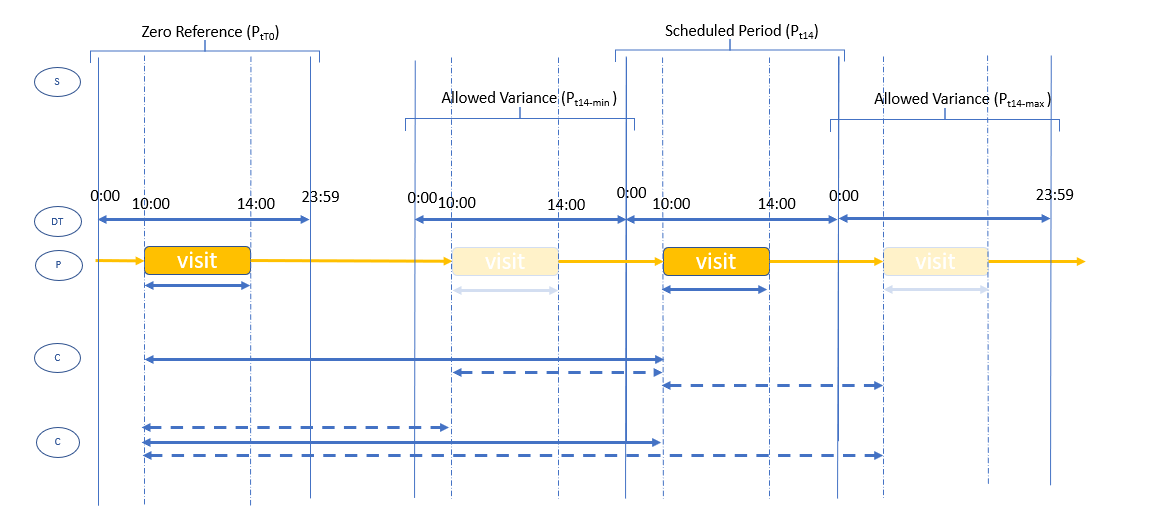
visit-window-research.png 
|
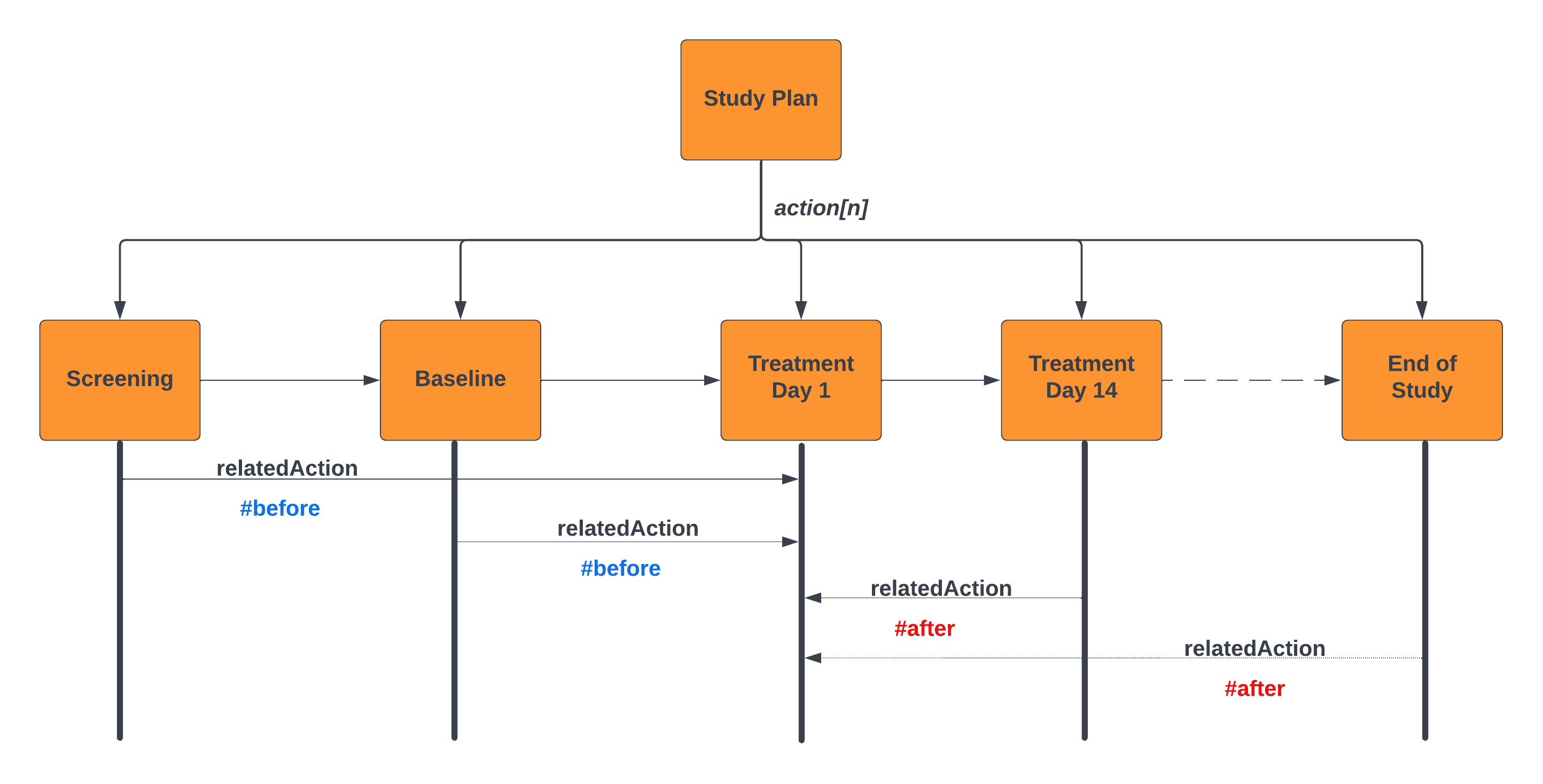
vulcan_soa_related_actions.png 
|