Clinical Study Schedule of Activities, published by HL7 International / Biomedical Research and Regulation. This guide is not an authorized publication; it is the continuous build for version 2.0.0-ballot built by the FHIR (HL7® FHIR® Standard) CI Build. This version is based on the current content of https://github.com/HL7/Vulcan-schedule-ig/ and changes regularly. See the Directory of published versions
| Page standards status: Informative |
Within the current implementation, it is possible to accommodate one or more schedules through use of the multiple entries in the ResearchStudy.protocol attribute based on the ResearchStudy resource
An illustration is shown here:
{
"resourceType": "ResearchStudy",
"id": "SampleMultiDesignStudy",
"title": "Sample Multi Design Study",
"protocol": [
{
"reference": "PlanDefinition/StudyDesignA"
},
{
"reference": "PlanDefinition/StudyDesignB"
}
]
}
Alternatively, a single study plan with conditional elements to represent all the points at which a multi-design protocol changes can be used. The designer will need to determine whether there needs to be separate protocol plan element, or whether a multi-design study should just be included within a single plan and use dynamic features to switch on and off parts of the study designs.
For an example of a master protocol, the existing ResearchStudy.partOf predicate can be used for the sub-studies as part of master protocol, this allows for differentiation of eligibility criteria, lifecycle, etc.
The modality of transitions are needed to represent a prospective plan for a ResearchSubject participating in a Clinical Trial; it supports planning and decision making. Generally, patients follow a protocol proscribed path through encounters and activities. We have previously described how activities within an encounter can be orchestrated; but this document is intended to summarise approaches for intra-encounter activities.
The designs should incorporate these directives in such a way that an application could interpret them to make decisions about the transitions; and thereby create the required resources (eg Encounter, Appointment, ServiceRequest). The challenge we have is that in CTMS systems, that are built around common conceptual understandings of how clinical trials work, the functions to drive these transitions are out of the box, whereas finding a common representation using FHIR resources may be challenging.
We have chosen to use the extensions proposed by Richardson A, Genyn P Clinical Trial Schedule of Activities Specification Using Fast Healthcare Interoperability Resources Definitional Resources: Mixed Methods Study JMIR Med Inform 2025;13:e71430 - henceforth referred to as MMS. The authors have kindly agreed for their work to be utilised as part of the IG, with the qualification that full recognition for the work shall remain theirs alone.
If we take a simple example; the progression of a patient in a study design - the following example provides an illustration
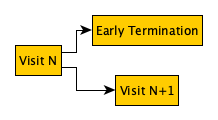
The following table represents a schedule of activities for this simple progression example:
| Visit N | Visit N+1 | Early Termination | |
|---|---|---|---|
| Study Day | 0 | 48 | Variable |
| Window | ±3d | ±3d | As needed |
| Activities | |||
| Informed Consent | ✓ | ||
| Physical Exam | ✓ | ✓ | ✓ |
| Vital Signs | ✓ | ✓ | ✓ |
| Laboratory Tests | ✓ | ✓ | ✓ |
| Study Drug Admin | ✓ | ||
| Adverse Events | ✓ | ✓ | ✓ |
| Concomitant Meds | ✓ | ✓ | ✓ |
| Discontinuation Reason | ✓ |
This simple design illustrates:
Here is a representation of this simple structure using the implementation details based on MMS:
{
"resourceType": "PlanDefinition",
"id": "dynamic-visit-schedule-simple-example",
"meta": {
"versionId": "0",
"lastUpdated": "2025-11-09T15:13:31Z"
},
"identifier": [
{
"system": "http://www.fhir4pharma.com/plandefinition",
"value": "5c2a9671-1d0d-4b02-8f09-0e30d77411b2"
}
],
"version": "V00",
"name": "dynamic-visit-schedule-simple-example",
"title": "dynamic-visit-schedule-simple-example",
"type": {
"coding": [
{
"code": "clinical-protocol",
"system": "http://terminology.hl7.org/CodeSystem/plan-definition-type"
}
]
},
"status": "active",
"publisher": "fhir4pharma [Richardson & Genyn, JMIR Med Inform 2025;13:e71430, DOI: 10.2196/71430]",
"description": "dynamic-visit-schedule-simple-example",
"action": [
{
"id": "ac4d0cb9-f2bd-49c1-8b28-42d5cd04b4fb",
"title": "Visit N",
"description": "Visit N",
"definitionCanonical": "http://example.org/Encounter/Visit-N",
"extension": [
{
"url": "http://fhir4pharma.com/StructureDefinition/soaPlannedTimepoint",
"extension": [
{
"url": "soaTimePointType",
"valueString": "Interaction"
},
{
"url": "soaPlannedTimePoint",
"valueQuantity": {
"value": 0,
"code": "d",
"system": "http://unitsofmeasure.org"
}
},
{
"url": "soaPlannedRange",
"valueRange": {
"low": {
"value": 0,
"code": "d",
"system": "http://unitsofmeasure.org"
},
"high": {
"value": 0,
"code": "d",
"system": "http://unitsofmeasure.org"
}
}
},
{
"url": "soaReferenceTimePoint",
"valueString": "Visit N"
},
{
"url": "soaRangeFromTimePoint",
"valueString": "Visit N"
},
{
"url": "soaPlannedDuration",
"valueDuration": {
"value": 24,
"code": "h",
"system": "http://unitsofmeasure.org"
}
},
{
"url": "soaRepeatAllowed",
"valueBoolean": false
}
]
}
],
"groupingBehavior": "visual-group",
"selectionBehavior": "exactly-one",
"action": [
{
"extension": [
{
"url": "http://fhir4pharma.com/StructureDefinition/soaTransition",
"extension": [
{
"url": "soaTargetId",
"valueString": "c25995f4-be76-47fa-ae90-a46100f8cfb3"
},
{
"url": "soaTransitionType",
"valueString": "FS"
},
{
"url": "soaTransitionDelay",
"valueDuration": {
"value": 48,
"code": "d",
"system": "http://unitsofmeasure.org"
}
},
{
"url": "soaTransitionRange",
"valueRange": {
"low": {
"value": 3,
"code": "d",
"system": "http://unitsofmeasure.org"
},
"high": {
"value": 3,
"code": "d",
"system": "http://unitsofmeasure.org"
}
}
}
]
}
],
"condition": [
{
"kind": "start",
"expression": {
"language": "text/x-soa-expressionplain",
"expression": "{'toNormalProgression':true}"
}
}
]
},
{
"extension": [
{
"url": "http://fhir4pharma.com/StructureDefinition/soaTransition",
"extension": [
{
"url": "soaTargetId",
"valueString": "349447c3-8ad4-4034-8c31-c3d96dcc5f9a"
},
{
"url": "soaTransitionType",
"valueString": "SS"
},
{
"url": "soaTransitionDelay",
"valueDuration": {
"value": 24,
"code": "h",
"system": "http://unitsofmeasure.org"
}
},
{
"url": "soaTransitionRange",
"valueRange": {
"low": {
"value": 0,
"code": "d",
"system": "http://unitsofmeasure.org"
},
"high": {
"value": 47,
"code": "d",
"system": "http://unitsofmeasure.org"
}
}
}
]
}
],
"condition": [
{
"kind": "start",
"expression": {
"language": "text/x-soa-expressionplain",
"expression": "{'toEarlyTermination':true}"
}
}
]
}
]
},
{
"id": "c25995f4-be76-47fa-ae90-a46100f8cfb3",
"title": "Visit N+1",
"description": "Visit N+1",
"definitionCanonical": "http://example.org/Encounter/Visit-N+1",
"extension": [
{
"url": "http://fhir4pharma.com/StructureDefinition/soaPlannedTimepoint",
"extension": [
{
"url": "soaTimePointType",
"valueString": "Interaction"
},
{
"url": "soaPlannedTimePoint",
"valueQuantity": {
"value": 48,
"code": "d",
"system": "http://unitsofmeasure.org"
}
},
{
"url": "soaPlannedRange",
"valueRange": {
"low": {
"value": 3,
"code": "d",
"system": "http://unitsofmeasure.org"
},
"high": {
"value": 3,
"code": "d",
"system": "http://unitsofmeasure.org"
}
}
},
{
"url": "soaReferenceTimePoint",
"valueString": "Visit N"
},
{
"url": "soaRangeFromTimePoint",
"valueString": "Visit N"
},
{
"url": "soaPlannedDuration",
"valueDuration": {
"value": 24,
"code": "h",
"system": "http://unitsofmeasure.org"
}
},
{
"url": "soaRepeatAllowed",
"valueBoolean": false
}
]
}
]
},
{
"id": "349447c3-8ad4-4034-8c31-c3d96dcc5f9a",
"title": "Early Termination",
"description": "Early Termination",
"definitionCanonical": "http://example.org/Encounter/Early-Termination",
"extension": [
{
"url": "http://fhir4pharma.com/StructureDefinition/soaPlannedTimepoint",
"extension": [
{
"url": "soaTimePointType",
"valueString": "Interaction"
},
{
"url": "soaPlannedTimePoint",
"valueQuantity": {
"value": 24,
"code": "h",
"system": "http://unitsofmeasure.org"
}
},
{
"url": "soaPlannedRange",
"valueRange": {
"low": {
"value": 0,
"code": "s",
"system": "http://unitsofmeasure.org"
},
"high": {
"value": 48,
"code": "d",
"system": "http://unitsofmeasure.org"
}
}
},
{
"url": "soaReferenceTimePoint",
"valueString": "Visit N"
},
{
"url": "soaRangeFromTimePoint",
"valueString": "Visit N"
},
{
"url": "soaPlannedDuration",
"valueDuration": {
"value": 24,
"code": "h",
"system": "http://unitsofmeasure.org"
}
},
{
"url": "soaRepeatAllowed",
"valueBoolean": false
}
]
}
]
}
]
}
The extension http://fhir4pharma.com/StructureDefinition/soaTransition is key to defining the paths forward;
PlanDefinition.action is assigned a unique identifier using id (this should be a UUID/GUID so as to ensure internal referential integrity)PlanDefinition.action has a child action for each of the possible outcomes;targetId represents the identifier of the target actionsoaTransitionType value represents the type of transition (eg Start to Start = SS)soaRepeatAllowed value defines whether it is possible to revisit this targetsoaPlannedDuration value specifies the duration at which this transition is expected to occurcondition attribute on the PlanDefinition.action.action is used to specify the 'logic' controlling the transition, and it follows the existing semantics of the action.condition.For a deeper discussion and definition of this, please refer to the source paper here: Clinical Trial Schedule of Activities Specification Using Fast Healthcare Interoperability Resources Definitional Resources: Mixed Methods Study.
In order to ensure that the transitions can be evaluable, for each action the evaluation of the condition should be a boolean, where true the transition should be allowed. Any action without a condition is the default transition, and should be presented if no other actions are applicable. A terminal activity will have no transitions available given all the conditions available.
The condition can be implemented using any of current languages (eg CQL, FHIRPath), but should also support feedback or input by a site coordinator. An example of where condition can be inferred from the healthcare system include a reference to the Subject milestones (egResearchSubject.status or ResearchSubject.subjectState/ResearchSubject.subjectMilestone), as discussed in dynamics - eg performing a particular activity is conditional on a patient having completed informed consent, perhaps for an experimental arm.
The execution of the plan needs to be able to be adapted to describe what transitions could occur and describe any conditions under which the transitions might occur; examples of the types of transitions that could need to be represented:
So, what needs to be defined for a given encounter forward in patient progression based on what activities are planned to occur next based on the protocol; some are common such as the Early Termination path; based on outcomes from the study (eg Serious Adverse Event, Lost to Follow-up), others can be be more complicated.
The following table represents the Schedule of Activities for a simple path example:
| Screening | Treatment | End of Study | Follow-up | |||
|---|---|---|---|---|---|---|
| Visit | Screen | Day 1 | Day 7 | Day 15 | EOS | FU |
| Study Day | -14 to -1 | 1 | 7 | 15 | 21 | +30 |
| Window | ±1d | ±2d | ±1d | ±7d | ||
| Activities | ||||||
| Informed Consent | ✓ | |||||
| Eligibility Assessment | ✓ | |||||
| Demographics | ✓ | |||||
| Medical History | ✓ | |||||
| Physical Exam | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Vital Signs | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Laboratory Tests | ||||||
| Hematology | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Chemistry Panel | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Treatment | ||||||
| Study Drug Admin | ✓ | |||||
| Assessments | ||||||
| Adverse Events | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Concomitant Meds | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Discontinuation Reason | ✓ | |||||
| Follow-up Assessment | ✓ |
This design illustrates:
This can be visualised as shown here:
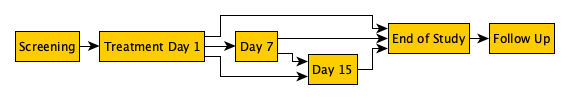
In each encounter there are two possible outcomes:
Using the extensions as defined, the representation of the study plan can be seen here
In this scenario, there is a design where the patients are randomised to one of two Arms; this example presents an asymmetric setup where the number of encounters differs for the two arms.
The following table represents the schedule of activities for this two-arm study design:
| Screening | Randomization | Treatment | End of Study | ||||
|---|---|---|---|---|---|---|---|
| Visit | Screen | Baseline | Day 1 | Day 21 | Day 7 | Day 15 | EOS |
| Study Day | -14 to -1 | 0 | 1 | 2 | 7 | 15 | 21 |
| Window | ±1d | ±1d | ±1d | ±2d | ±3d | ||
| Activities | |||||||
| Informed Consent | ✓ | ||||||
| Eligibility Assessment | ✓ | ||||||
| Demographics | ✓ | ||||||
| Medical History | ✓ | ||||||
| Randomization | ✓ | ||||||
| Physical Exam | ✓ | ✓ | ✓ | ✓1 | ✓ | ✓ | ✓ |
| Vital Signs | ✓ | ✓ | ✓ | ✓1 | ✓ | ✓ | ✓ |
| Laboratory Tests | |||||||
| Hematology | ✓ | ✓ | ✓ | ✓1 | ✓ | ✓ | ✓ |
| Chemistry Panel | ✓ | ✓ | ✓ | ✓1 | ✓ | ✓ | ✓ |
| Treatment | |||||||
| Drug A Admin2 | ✓ | ✓ | |||||
| Drug B Admin3 | ✓ | ||||||
| Assessments | |||||||
| Adverse Events | ✓ | ✓ | ✓ | ✓1 | ✓ | ✓ | ✓ |
| Concomitant Meds | ✓ | ✓ | ✓ | ✓1 | ✓ | ✓ | ✓ |
| Study Completion | ✓ |
1 Arm A only
2 Arm A only - administered on Days 1 and 2
3 Arm B only - administered on Day 1 only
This two-arm design illustrates:
This provides a visual representation of the encounters/transitions involved in the study design.
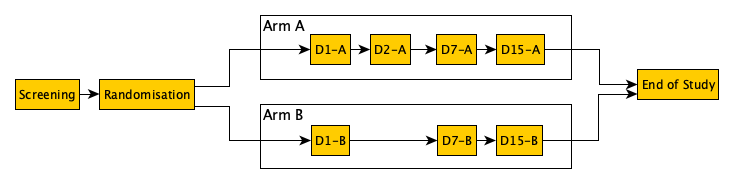
Note; the decision made for randomization should only need to be done once; once a patient is following the path for the assigned arm, the decision support system should preclude the other path (while retaining the common exit paths). The design should support 'common' planned encounters that can be used both before and after randomization. The nature of the FHIR resources and relationships between them should be able to be used to be most efficient.
The representation of this is shown here
One of the more complex scenarios we need to deal with are treatment cycles; these are repeatable episodic sets of encounters, usually in oncology studies. The structure of cycles may change over the progression of the studies; different encounters or activities occur based on the protocol. The status of the patient is evaluated at the end of each cycle, usually against a standard criteria like RECIST. The progression of the disease is the key determinant to whether the patient will continue to stay in the study (alongside all other safety measures discussed).
Some examples of Oncology Cycles:
The following illustrates an example oncology study design with repeating treatment cycles that have different visit patterns for even and odd cycles. The diagram shows cycles as distinct entities with clear transitions between cycles and within cycles:
| Screening | Cycle N (Odd) | Cycle N+1 (Even) | … | End of Treatment | Follow-up | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Visit | Screen | Rand | D1 | D14 | D28 | D1 | D7 | D14 | D21 | D28 | … | EOT | FU1 | FU2 |
| Study Day | -28 to -1 | 0 | 1 | 14 | 28 | 29 | 35 | 42 | 49 | 56 | … | +30 | +90 | +180 |
| Window | ±2d | ±2d | ±2d | ±2d | ±2d | ±2d | ±2d | ±2d | ±2d | ±7d | ±14d | ±14d | ||
| Activities | ||||||||||||||
| Informed Consent | ✓ | |||||||||||||
| Demographics | ✓ | |||||||||||||
| Medical History | ✓ | |||||||||||||
| Physical Exam | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Vital Signs1 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Height/Weight | ✓ | ✓ | ✓ | ✓ | ✓ | |||||||||
| Performance Status | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ||||
| Laboratory Tests | ||||||||||||||
| Hematology | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Chemistry Panel | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Liver Function | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Coagulation | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ||||||||
| Biomarkers | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |||||
| Treatment | ||||||||||||||
| Study Drug Admin | ✓ | ✓ | ✓ | |||||||||||
| Pre-medication | ✓ | ✓ | ✓ | |||||||||||
| Imaging | ||||||||||||||
| CT/MRI Scan | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ||||||||
| Chest X-ray | ✓ | ✓ | ✓ | |||||||||||
| Disease Response | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ||||||||
| Assessments | ||||||||||||||
| Quality of Life | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ||||||
| Adverse Events | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Concomitant Meds | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
In this design we pivot between odd-numbered cycles (Cycle 1, 3, 5) and even-numbered cycles (Cycle 2, 4, 6); we have assigned a disease response activity at the end of the even numbered cycles - these could be aligned to any of the usual grading schemes (eg RECIST), with the outcome of the assessment feeding into the decision over continuing in treatment cycles or transitioning to end of Treatment/follow up.
This can be illustrated graphically as follows:
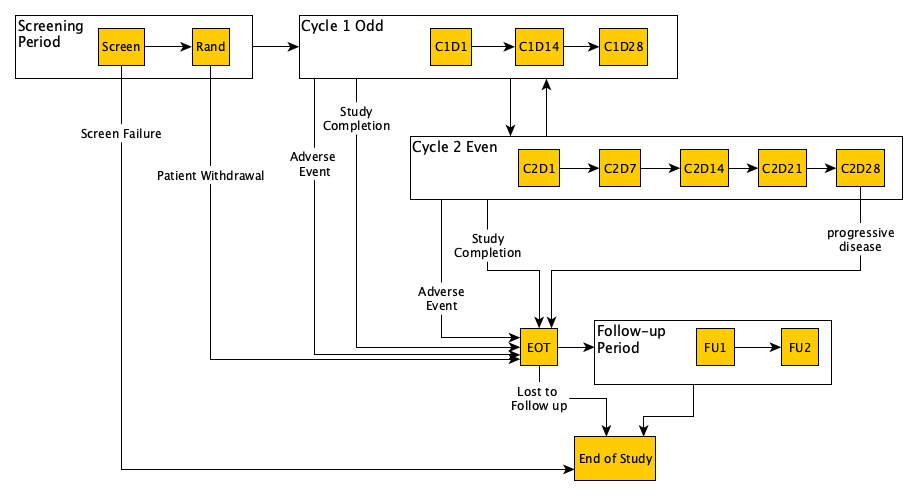
The representation of this is shown here.
Using the extensions we can represent the SoA as a Graph - this is key to being able to provide decision support in a prospective manner for a user utilising a healthcare system to manage the execution of a clinical trial. There are analogues to the implementation of Timelines in the CDISC Unified Study Design Model (USDM); so transformations between representations of Study Designs should be mappable between the standards.
The complexity of the FHIR study plan will be concordant with the complexity of the study design. The bias should always be towards implementation; working through an a