Lithuanian Breast Diagnostics Implementation Guide
0.0.1 - ci-build
Lithuanian Breast Diagnostics Implementation Guide
0.0.1 - ci-build
Lithuanian Breast Diagnostics Implementation Guide, published by Lithuanian Medical Library. This guide is not an authorized publication; it is the continuous build for version 0.0.1 built by the FHIR (HL7® FHIR® Standard) CI Build. This version is based on the current content of https://github.com/HL7LT/ig-lt-breast/ and changes regularly. See the Directory of published versions
Lithuanian
Language: en
| LinkID | Text | Definition | Answer |
|---|---|---|---|
 | Questionnaire:Questionnaire: Pathological Histological Examination ESPBI Form (LT Breast) | ||
  | Referral to perform pathology test | ||
  | Patient information | ||
   | Personal code | 48501019999 | |
   | Name | Janette | |
   | Surname | Petrauskaite | |
   | Date of birth | 1985-01-01 | |
   | Age | 40 | |
   | Gender | AdministrativeGender: female (Female) | |
  | Specimen dates | ||
   | Date of material collection | 2025-03-07T09:15:00+02:00 | |
   | Shipment / formation date | 2025-03-07T10:00:00+02:00 | |
  | Clinical diagnosis | ||
   | Clinical diagnosis ICD-10-AM code | C50.4 | |
   | Clinical diagnosis name | Malignant neoplasm of upper-outer quadrant of breast | |
  | Material fixation | ||
   | Material fixation type | Breast questionnaire answer options (illustrative): fixation-formalin (Fixed (10% formalin buffered solution)) | |
  | Previous procedures and key clinical data | ||
   | Key clinical data | Breast questionnaire answer options (illustrative): proc-none (No important information) | |
   | Other information | ||
  | Special tests | ||
   | Special tests required | IHC panel (ER, PR, HER2, Ki67) | |
  | Radiological examination data | ||
   | Radiological examination performed | Breast questionnaire answer options (illustrative): yes (Yes) | |
   | Examination type | ||
   | Examination date | 2025-03-05 | |
   | BI-RADS right breast | Breast questionnaire answer options (illustrative): birads-1 (BI-RADS 1 – No changes found) | |
   | BI-RADS left breast | Breast questionnaire answer options (illustrative): birads-4 (BI-RADS 4 – Suspicious malignant finding) | |
  | Operation details | ||
   | Nature of operation | Breast questionnaire answer options (illustrative): op-thick-needle-biopsy (Thick needle biopsy) | |
   | Other operation details | ||
  | Description of histological sample / object | ||
   | Container information | 1 container, labelled 'Left breast UOQ, CNB' | |
   | Biopsy location | Left breast, upper-outer quadrant, 2 o'clock position, 5 cm from nipple | |
  | Description of macroscopic examination | ||
   | Biopsy size (mm) | 15 mm (Details: UCUM codemm = 'mm') | |
   | Fragment details | 4 tissue cores, pale grey, firm, longest 15 mm | |
   | Notes | Entirely submitted in 2 cassettes | |
  | Description of microscopic examination | ||
   | Microscopic examination description | Core biopsy fragments showing invasive carcinoma of no special type (NST). Tumour cells arranged in nests, cords and tubular structures, surrounded by desmoplastic stroma. Moderate nuclear pleomorphism, mitotic count 8/10 HPF. No definite in-situ component identified in this limited sample. | |
  | Immunohistochemistry (IHC) | ||
   | Estrogen receptors ER | Breast questionnaire answer options (illustrative): ihc-strong (Strong (+++)) | |
   | ER expression in nuclei (%) | 90 % (Details: UCUM code% = '%') | |
   | Progesterone receptors PR | Breast questionnaire answer options (illustrative): ihc-moderate (Moderate (++)) | |
   | PR expression in nuclei (%) | 60 % (Details: UCUM code% = '%') | |
   | HER2 status | Breast questionnaire answer options (illustrative): her2-borderline (HER2: expression borderline (2+)) | |
   | HER2 evaluation | Breast questionnaire answer options (illustrative): her2-eval-2 (HER2 evaluation: 2+) | |
  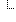 | Ki67 proliferation index (%) | 25 % (Details: UCUM code% = '%') | |
  | Molecular studies | ||
   | FISH result | Breast questionnaire answer options (illustrative): fish-not-evaluated (FISH: not to be evaluated) | |
   | HER2 average | ||
   | Average CEP17 | ||
   | HER2/CEP17 ratio | ||
   | CISH/SISH result | Breast questionnaire answer options (illustrative): cish-not-evaluated (CISH/SISH: not to be evaluated) | |
  | Conclusion | ||
   | WHO Classification diagnosis | Invasive breast carcinoma of no special type (NST) (ICD-O 8500/3) | |
   | Tumour grade | Breast questionnaire answer options (illustrative): grade-g2 (G2 (moderately differentiated)) | |
   | Tumour size (mm) | 18 mm (Details: UCUM codemm = 'mm') | |
   | Lymphovascular invasion | Breast questionnaire answer options (illustrative): lvi-0 (pLVI-0: not detected) | |