US Prescription Drug Monitoring Program (PDMP), published by HL7 International / Pharmacy. This guide is not an authorized publication; it is the continuous build for version 1.0.0-ballot built by the FHIR (HL7® FHIR® Standard) CI Build. This version is based on the current content of https://github.com/HL7/fhir-pdmp/ and changes regularly. See the Directory of published versions
Prescription Drug Monitoring Programs (PDMPs) are state-based databases that provide prescribers and pharmacists with timely information about controlled substance, and in some states non-controlled substance, dispensing, administration and patient behaviors. Use of information stored in PDMPs during care delivery helps avoid drug misuse and diversion and can provide improved patient care and safety.
To reduce opioid misuse, reduce drug diversion, and for other purposes, states have implemented policies mandating providers to reference PDMPs to obtain a patient’s PDMP history before prescribing or dispensing certain medications. The Prescription Drug Monitoring Program (PDMP) FHIR Implementation Guide defines a method for providers to request and retrieve patient PDMP information using the HL7 FHIR standard.
For general background on state PDMP programs, see the Centers for Disease Control and Prevention PDMP - What States Need to Know.
The Figure 1, below, shows an overview of the PDMP reporting ecosystem. This diagram was adapted from Prescription Drug Monitoring Program Training and Technical Assistance Center [PDMP TTAC(https://www.pdmpassist.org)]. PDMP TTAC is an organization of PDMPs, federal partners and other stakeholders which has establish technical standards and provides other services, support, resources, & strategies to further the efforts and effectiveness of PDMPs.
In this figure,
Outside of a user accessing a State PDMP System via a web portal, the interactions in Figure 1 can support discrete data exchange.
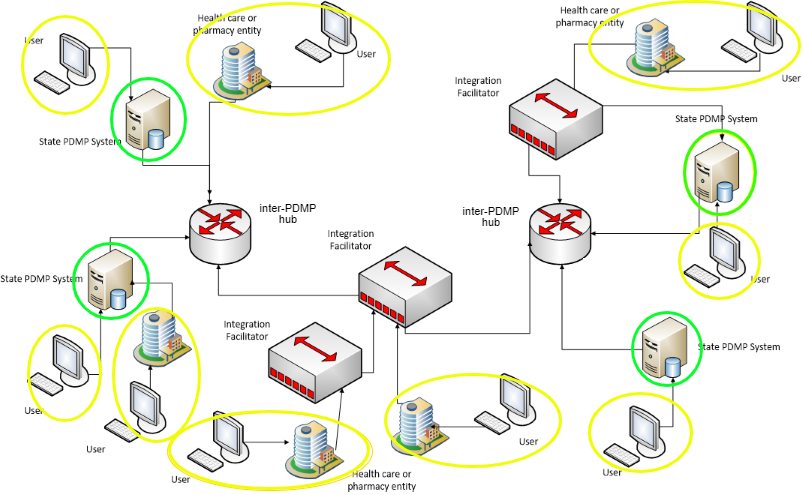
This Implementation Guide provides another method for Users (or Users + Health care or Pharmacy entities) to request and receive PDMP history reports. Systems may elect to follow the method in this Implementation Guide to leverage existing FHIR Infrastructure, particularly when discrete PDMP data is allowed to be stored or incorporated locally.
For this Implementation Guide, we limit and abstract the ecosystem in Figure 1 to focus on PDMP request and PDMP response messages exchanged between the PDMP Requester and the PDMP Responder as depicted in Figure 2.
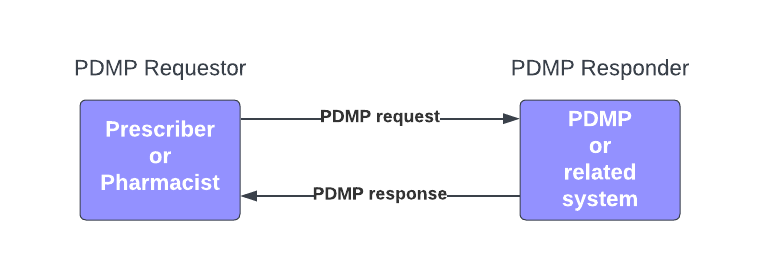
To support varying regulatory requirements, PDMP response supports both the PDMP history as discrete data and a URL pointing to the PDMP history rendered (fully-formatted) to regulatory requirements.
This guide provides both RESTful operation and messaging submission methods to match implementers’ particular environments as described here.
This Implementation Guide is intended to be used in the United States. It reflects US pharmacy processes and conventions.
Hubs and Integration Facilitators The inter-PDMP hubs and Integration Facilitators in Figure 1 may be associated with either the PDMP Requestor or the PDMP Responder in Figure 2, depending upon the implementation. They are not specifically addressed as separate entities in this Implementation Guide.
Other interactions between PDMP ecosystem parties, including parties not depicted in Figure 1, are out of scope for this Implementation Guide. These exchanges include, but are not limited to:
Patient Matching: This Implementation Guide does not specify patient matching requirements. States may require requesters to include certain patient information to enable or facilitate patient matching. This guide supports that patient information, however, the specific patient matching criteria requirements for each PDMP are out of scope.
PDMP Logging Requirements: This Implementation Guide does not address PDMP logging requirements since the requirements vary between jurisdictions and are not typically messaged (i.e., not communicated back to the PDMP).
PDMP Requester: A system that sends PDMP requests to PDMP Responders and receives PDMP responses in return. The system may include both a local system (e.g., EHR, Pharmacy System) and associated intermediaries (e.g., message routing and other value-added service providers). This system can be thought of as the client in a client-server interaction.
PDMP Responder: A system that receives PDMP requests from PDMP Requesters and responds with PDMP responses. The system may include, in addition to the state PDMP, other partner systems (e.g., hubs). This system can be thought of as the server in a client-server interaction.
PDMP request: A message, sent by a PDMP requester on behalf of a user, which contains all necessary and required information such that a PDMP Responder can, if appropriate, respond with a PDMP response. This includes, but is not limited to, user identification and authorization and patient identification.
PDMP response: A message, sent by a PDMP Responder that particular PDMP Requester that submitted a specific PDMP request. The message may include any combination of: the PDMP history in discrete data, a URL to a formatted PDMP history report, additional alerts and message, error messages, and other relevant information.
PDMP history: The content of a PDMP response including pertinent patient, pharmacy, prescriber, and dispensation records. The PDMP Responder may also include alerts, administration records, and other information as appropriate or as required by policy or regulation.
The guide is organized into the following sections:
For those new to FHIR, the material below describes basic FHIR principles and gives guidance for reading FHIR specifications.
| HL7 Pharmacy Workgroup | |
| Frank McKinney | frank.mckinney@pocp.com |
| Scott Robertson | scott.robertson@pocp.com |
| Brett Marquard, Nagesh Bashyam, Melva Peters and Eric Haas authored the predecessor to this guide, the US Meds FHIR IG. Certain content from that work has been updated and included here. | |
The authors acknowledge and thank our stakeholders for their essential contribution. A limited number of those stakeholders are listed here:
| Carly Crownover | Database Administrator | CSMD |
| Craig Wetzelberger | Director of Software Engineering | Bamboo Health |
| Dara Zarley, PharmD | Grant & Project Analyst | Nevada State Board of Pharmacy |
| Don Vogt | Prescription Drug Monitoring Program Training and Technical Assistance Center (PDMP TTAC) | |
| Fan Xiong, MPH | Senior Epidemiologist | Washington State Department of Health |
| Fred Aabedi | CTO | LogiCoy Inc |
| Jean Hall | Program Director, PDMP Solutions | LogiCoy Inc. |
| Jim Giglio | Prescription Drug Monitoring Program Training and Technical Assistance Center (PDMP TTAC) | |
| Joseph Fibich, PharmD | Director, Pharmacy Services Program | CyncHealth |
| Keller Martin | Staff Solutions Architect | Bamboo Health |
| Kevin Borcher | Prescription Drug Monitoring Program Training and Technical Assistance Center (PDMP TTAC) | |
| Mitchell Barnett, PharmD, MS | Associate PMP Director | Iowa PDMP |
| Nerissa Montgomery | Louisiana Board of Pharmacy | |
| Pat Knue | Prescription Drug Monitoring Program Training and Technical Assistance Center (PDMP TTAC) | |
| Rachel Buelow | Surescripts | |
| Reshmi Ravindranath | PMP Administrator | Louisiana Board of Pharmacy |
| Robert May | Program Director | IJIS Institute |
| Ronald Larsen | PDMP Consultant | IJIS Institute - RxCheck |
| Siri Chadalavada | Biztek Innovations | |
| Wanda Govan-Jenkins, DNP, MBA, MS, RN | ONC Project Officer Lead | Office of the National Coordinator for Health Information Technology |
| Zohaib Salim | Senior Product Manager | Bamboo Health |
| IG | Package | FHIR | Comment |
|---|---|---|---|
| hl7.fhir.us.pdmp#1.0.0-ballot | R4 | ||
| hl7.terminology.r4#5.5.0 | R4 | Automatically added as a dependency - all IGs depend on HL7 Terminology | |
| hl7.fhir.uv.extensions.r4#5.1.0 | R4 | Automatically added as a dependency - all IGs depend on the HL7 Extension Pack | |
| hl7.fhir.us.core#6.1.0 | R4 | ||
| hl7.terminology.r4#5.0.0 | R4 | ||
| hl7.fhir.uv.extensions.r4#1.0.0 | R4 | ||
| hl7.fhir.uv.bulkdata#2.0.0 | R4 | ||
| hl7.fhir.uv.smart-app-launch#2.1.0 | R4 | ||
| us.nlm.vsac#0.11.0 | R4 | ||
| hl7.fhir.uv.sdc#3.0.0 | R4 | ||
| hl7.fhir.r4.examples#4.0.1 | R4 | ||
| us.cdc.phinvads#0.12.0 | R4 | ||
| ihe.formatcode.fhir#1.1.0 | R4 |
Package hl7.fhir.uv.extensions.r4#5.1.0 This IG defines the global extensions - the ones defined for everyone. These extensions are always in scope wherever FHIR is being used (built Sat, Apr 27, 2024 18:39+1000+10:00) |
Package hl7.fhir.uv.extensions.r4#1.0.0 This IG defines the global extensions - the ones defined for everyone. These extensions are always in scope wherever FHIR is being used (built Sun, Mar 26, 2023 08:46+1100+11:00) |
Package hl7.fhir.uv.bulkdata#2.0.0 FHIR based approach for exporting large data sets from a FHIR server to a client application (built Fri, Nov 26, 2021 05:56+1100+11:00) |
Package hl7.fhir.r4.examples#4.0.1 Example resources in the R4 version of the FHIR standard |
Package hl7.fhir.uv.sdc#3.0.0 The SDC specification provides an infrastructure to standardize the capture and expanded use of patient-level data collected within an EHR. |
Package ihe.formatcode.fhir#1.1.0 Implementation Guide for IHE defined FormatCode vocabulary. (built Thu, Feb 24, 2022 16:55-0600-06:00) |
Package hl7.fhir.us.core#6.1.0 The US Core Implementation Guide is based on FHIR Version R4 and defines the minimum conformance requirements for accessing patient data. The Argonaut pilot implementations, ONC 2015 Edition Common Clinical Data Set (CCDS), and ONC U.S. Core Data for Interoperability (USCDI) v1 provided the requirements for this guide. The prior Argonaut search and vocabulary requirements, based on FHIR DSTU2, are updated in this guide to support FHIR Version R4. This guide was used as the basis for further testing and guidance by the Argonaut Project Team to provide additional content and guidance specific to Data Query Access for purpose of ONC Certification testing. These profiles are the foundation for future US Realm FHIR implementation guides. In addition to Argonaut, they are used by DAF-Research, QI-Core, and CIMI. Under the guidance of HL7 and the HL7 US Realm Steering Committee, the content will expand in future versions to meet the needs specific to the US Realm. These requirements were originally developed, balloted, and published in FHIR DSTU2 as part of the Office of the National Coordinator for Health Information Technology (ONC) sponsored Data Access Framework (DAF) project. For more information on how DAF became US Core see the US Core change notes. (built Fri, Jun 30, 2023 14:02+0000+00:00) |
This is an R4 IG. None of the features it uses are changed in R4B, so it can be used as is with R4B systems. Packages for both R4 (hl7.fhir.us.pdmp.r4) and R4B (hl7.fhir.us.pdmp.r4b) are available.
There are no Global profiles defined
This publication includes IP covered under the following statements.